In this issue of Blood, Nair et al report on a computationally derived hepatocyte-specific cis-regulatory module (HS-CRM8) to accomplish high tissue-specific expression in gene therapy for hemophilia. When HS-CRM8 is introduced upstream of a minimal liver-specific promoter in an adenoassociated virus (AAV) that expresses a codon-optimized hyperactive human factor IX (FIX) mutant (FIX Padua), it provides a >1 log increase in systemic FIX protein levels and supraphysiological activity over a range of vector doses.1
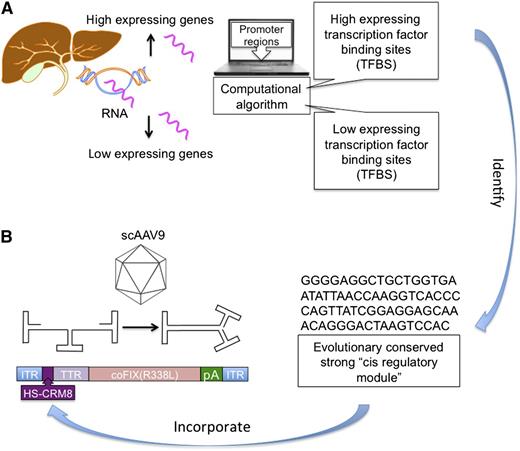
Schematic of the computational approach for identifying an HS-CRM8. (A) Overview of steps involved in HS-CRM8 identification. Human liver tissue was used to identify genes that are specifically highly or lowly expressed by using microarrays. Next, the respective promoters for the highly and lowly expressed genes were extracted from existing databases. Transcription factor binding sites (TFBSs) were then annotated by using the TRANSFAC database and were subjected to a computational algorithm to identify HS-CRM8, which was further refined by analysis of evolutionary conservation. (B) Schematic of the best performing scAAV9 vector used in the study with a codon-optimized human factor IX Padua mutant (cohFIX R338L) expressed from the HS-CRM8 and a minimal transthyretin (TTR) promoter.
Schematic of the computational approach for identifying an HS-CRM8. (A) Overview of steps involved in HS-CRM8 identification. Human liver tissue was used to identify genes that are specifically highly or lowly expressed by using microarrays. Next, the respective promoters for the highly and lowly expressed genes were extracted from existing databases. Transcription factor binding sites (TFBSs) were then annotated by using the TRANSFAC database and were subjected to a computational algorithm to identify HS-CRM8, which was further refined by analysis of evolutionary conservation. (B) Schematic of the best performing scAAV9 vector used in the study with a codon-optimized human factor IX Padua mutant (cohFIX R338L) expressed from the HS-CRM8 and a minimal transthyretin (TTR) promoter.
Currently, patients with hemophilia B, an X-linked coagulation disorder resulting from loss of functional FIX protein, are being recruited in 3 clinical trials evaluating different AAV-FIX vectors (serotype 8) for liver-directed gene transfer. However, it has now become clear from multiple clinical studies that vector doses above a threshold level provoke anti-capsid immunity and can limit long-term therapeutic gene expression.2 Although transient immune suppression protocols are evolving to minimize loss of therapeutic gene expression, most protocols are reactive, thus risking at least partial elimination of transgene-expressing tissues by capsid-specific CD8+ T cells. Therefore, developing AAV vectors that can avoid immune sensing and elimination from target tissues would have a significant impact on the long-term success of clinical studies of gene therapy. It was initially hoped that using alternate serotypes with high liver tropism would be a straightforward solution to the problem. So far, this approach has not translated from mice to humans, but it has prompted studies on selection of capsids with superior transduction of human hepatocytes in vivo using mice with humanized livers.3 In the meantime, research has focused mainly on these 3 approaches: (1) developing AAV vectors with stealthier capsids that have reduced presentation of major histocompatibility complex class I; (2) limiting innate immune signals; and (3) improving FIX expression levels and specific activity, with the goal of obtaining therapeutic FIX expression while avoiding detection by the immune system.4-7
By using a computational approach, Nair et al identified highly and lowly expressed genes in human liver tissue. After eliminating lowly expressed genes, they extracted the highly expressing gene promoter sequences and transcription factor binding sites from a database and processed the data through an analysis algorithm (panel A). The result was a highly conserved CRM within the alpha-1-antitrypsin promoter HS-CRM8. To stringently test the specificity of their HS-CRM8, they investigated transgene expression from an AAV serotype 9 (AAV9) vector, which transduces a variety of tissues in mice (panel B). Transgene expression was detected only in the liver, demonstrating the desired restricted expression. Importantly, the authors were able to restore hemostasis in mice with hemophilia B at the extremely low vector dose of 5 × 1010 vector genomes per kilogram (vg/kg). To put this into context, patients with hemophilia B have been dosed in the range of 2 × 1011 to 2 × 1012 vg/kg. One of the most remarkable findings of this study is the identification of the HS-CRM8 that enhances liver-specific gene expression although it is only 71 bp long. To understand the significance of this finding, one must consider the biology of AAV vectors. One of the main limitations of AAV vectors is that viral particles can package approximately 4.5 kb of DNA, including regulatory elements, introns, therapeutic transgenes, and polyadenylation signals. Self-complementary AAV (scAAV) vectors that are used in 2 ongoing clinical trials for hemophilia B because they do not require second-strand synthesis and thus show enhanced transduction, can package only half as much (<2.5 kb). Hence, these scAAV vectors can especially benefit from small yet powerful transcriptional regulatory elements. If the HS-CRM8 performs similarly in humans with the codon-optimized Padua mutant of FIX, it could provide an increase of up to 100-fold in FIX activity over the vectors currently being evaluated in clinical trials. Such an increase might allow for a reduction in vector dose below the threshold that activates anti-capsid immune responses, while reaching near physiological activity levels. The ability to express high antigen levels at low vector doses may also facilitate development of gene therapy–based immune tolerance protocols for reversal of inhibitor formation in hemophilic patients because the success of this strategy is dependent on expression level.8 One caveat that will need to be addressed in future studies is that neutralizing antibodies to AAV capsid will be more effective at reduced vector doses.6
The HS-CRM8 element might also have a major impact on gene therapy for hemophilia A. Obtaining high levels of factor VIII expression from an AAV vector has been challenging because of the large size of the complementary DNA (cDNA) (∼4.3 kb), which is nearly the packaging capacity of a single-stranded AAV vector. Although codon optimization has been helpful, a small element that is superior to current promoters is desirable.9,10 The same conclusion applies to AAV vectors (and other vector systems) encoding large therapeutic cDNAs that are in development for other diseases. Moreover, the method of in silico analysis of regulatory sequences for identification of optimal tissue-specific and/or cell-specific enhancer combinations should be broadly applicable to the optimization of therapeutic gene expression in any target tissue, including skeletal and cardiac muscles, the central nervous system and/or neurons, vascular endothelial cells, lung tissue, and others. Going forward with hepatic gene transfer or other targets, evaluation in large animal models, including non-human primates, is desirable. Regardless, the work by Nair et al shows that multiple elements (scAAV, codon-optimized cDNA, hyperactivity mutation, and a computationally derived regulatory module) can be combined to work synergistically to generate a more effective gene therapy vector for the treatment of hemophilia.
Conflict-of-interest disclosure: R.W.H. has been receiving royalty payment from Genzyme Corp. for license of AAV-FIX technology. The remaining author declares no competing financial interests.