To the editor:
Hepatitis E virus (HEV) is a small nonenveloped RNA virus usually transmitted by the enteric route, although transmission by blood transfusion has also been reported.1-3 HEV infection usually leads to benign acute hepatitis. It can sometimes be fulminant, particularly among pregnant women and patients with preexisting liver disease, or evolve to a chronic state, especially in immunosuppressed subjects.1,4
Pathogen reduction (PR) of blood products (BPs) has demonstrated its effectiveness with regard to a large number of pathogens.5,6 Among PR methods, the Intercept method combines a synthetic psoralene amotosalen HCl treatment with ultraviolet A (UVA) light illumination to block the replication of DNA and RNA.
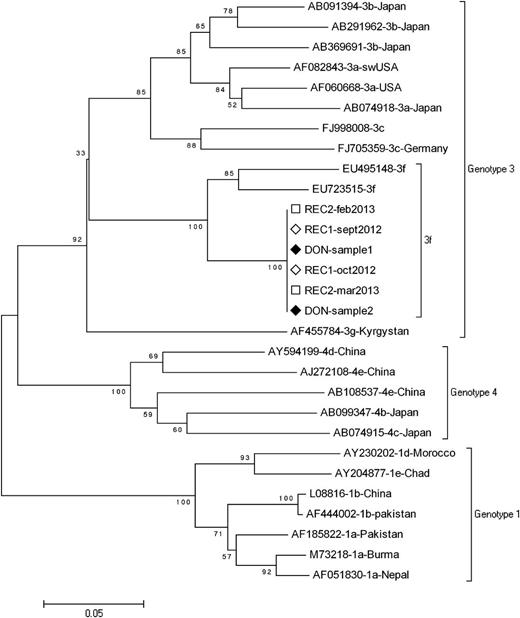
We report 2 cases of HEV transmission by 2 units of Intercept-treated plasma originating from the same donor. The first patient is a 36-year-old man with chronic renal failure. He underwent a kidney transplantation, which was followed by acute humoral rejection and was treated by plasma exchanges from March/June 2012, during which 59 BPs were transfused. Liver cytolysis was observed since June 2012. The diagnosis of hepatitis E was reached in October 2012 with detectable HEV RNA and weakly reactive anti-HEV immunoglobulin M. As of June 2013, the patient remained viremic, and ribavirin was introduced. HEV RNA was undetectable on the day of transplantation, as well as on the graft donor, but was detected in apheresis donation leading to transfused Intercept-treated fresh frozen plasma (FFP). The second patient is a 61-year-old man who underwent a liver transplantation for alcoholic liver cirrhosis in August 2012. Hepatitis E infection was detected in February 2013 with detectable HEV RNA and negative HEV serology. As of April 2013, the patient remained viremic, and ribavirin was introduced. He had received 72 BPs; HEV RNA was undetectable on the day of transplantation and in the graft donor but was detected in apheresis donation leading to transfused Intercept-treated FFP. All other blood donations for these patients tested negative for HEV RNA (using cryopreserved plasma samples collected on donation day). Further investigations revealed that the incriminated FFPs resulted from the same apheresis donation that was amotosalen/UVA light treated before segmentation in 3 units. Two of the 3 units were transfused to the 2 patients described above; the third was transfused to a patient who died 2 days following transfusion. The 2 FFP recipients and the donor were infected by a genotype 3f strain presenting a strict homology on partial sequences of the open reading frame 1 (ORF1) and ORF2 regions as previously described (Figure 1).7 Such strain identity demonstrates that both amotosalen and UVA light-treated FFPs provided by a unique donor transmitted HEV to ≥2 transfusion recipients. The involved donor was a 32-year-old woman who did not reveal any factor that could suggest that she was infected by HEV during the period of donation.
Phylogenetic analysis of partial ORF1 sequence. Phylogenetic tree was constructed on the MEGA4 software using the neighbor-joining method from a Kimura 2-parameter distance matrix based on partial nucleotide sequences of ORF1 encoding for RdRp, as previously described (4). Bootstrap values obtained from 500 resamplings are shown. A 100% homology is observed between blood donor sequences obtained from 2 aliquots of the same donation (DON sample 1 and sample 2) and recipient’s sequences (REC1 and REC2) obtained from 2 different samples. GenBank reference sequences are indicated by their accession number.
Phylogenetic analysis of partial ORF1 sequence. Phylogenetic tree was constructed on the MEGA4 software using the neighbor-joining method from a Kimura 2-parameter distance matrix based on partial nucleotide sequences of ORF1 encoding for RdRp, as previously described (4). Bootstrap values obtained from 500 resamplings are shown. A 100% homology is observed between blood donor sequences obtained from 2 aliquots of the same donation (DON sample 1 and sample 2) and recipient’s sequences (REC1 and REC2) obtained from 2 different samples. GenBank reference sequences are indicated by their accession number.
Such novel HEV transmission through Intercept-treated FFP establishes resistance of HEV to Intercept PR technology. Nonenveloped viruses such as HEV are also known to be resistant to solvent/detergent treatment. In vitro, assessment has established that hepatitis A virus, a similar nonenveloped virus, as well as feline calicivirus, a model for HEV, are poorly sensitive to amotosalen/UVA light.5,6 Hepatitis E has recently emerged as a significant cause of transfusion-induced viral hepatitis. Generalizing HEV screening for all blood donations, or alternatively for a fraction of BPs to be transfused in high-risk patients, is being considered in France.
Authorship
Acknowledgments: L.H. designed research, collected, analyzed and interpreted data, and wrote the manuscript; A.-M.R.-A. performed biological analysis and wrote the manuscript; A.B., M.S., and N.B.d.R. collected data, participated to the patients' diagnosis; B.D.F. and V.M. diagnosed the disease and followed the first patient; P.T. interpreted data and provided critical review of the manuscript; and P.B. designed research, interpreted data, wrote and edited the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Philippe Bierling, Etablissement Français du Sang Ile de France, 122-130 rue Marcel Hartmann, Léa Park, Bat B, 94200 Ivry sur Seine, France; e-mail: philippe.bierling@efs.sante.fr.