Abstract
FcγRIIA-mediated platelet activation is important in the pathogenesis of heparin-induced thrombocytopenia (HIT) and other immune-mediated thrombocytopenia and thrombosis disorders. FcgRIIa is also the major transmembrane signaling adapter for the human platelet fibrinogen receptor, integrin aIIbb3. In HIT, antibody to PF4/heparin is necessary but not sufficient for the disease. There is considerable inter-individual variation in platelet FcγRIIA activation; the reasons remain unclear. To identify potential regulators in the FcγRIIA pathway, we determined the platelet RNA expression profile in 154 healthy human donors who were phenotyped for platelet aggregation via FcgRIIA. 87 mRNAs and 4 DE miRNAs were identified as differentially expressed (DE) between hyper- and hypo-responsive donors by microarray and Nanostring analysis, respectively. Among them, hyper-responders show significantly lower T-cell ubiquitin ligand-2 (TULA-2) mRNA level and higher miR-148a-3p and miR-25-3p levels than hyporesponders. TULA-2, also known as UBASH3B, is a known protein tyrosine phosphatase for Syk, and thus a biologically plausible regulator of platelet FcgRIIA.
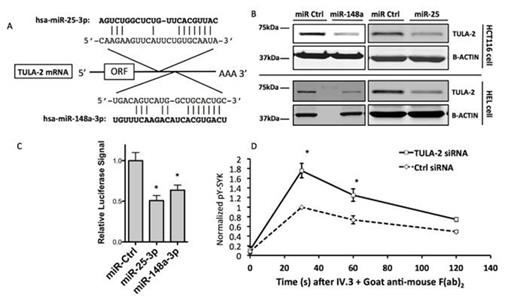
Both miR-148a-3p and miR-25-3p are predicted to target the 3’UTR region on TULA-2 mRNA (Fig., A). Overexpression of miR-148a-3p or miR-25-3p results in down-regulation of the TULA-2 protein level in HCT116 cells and HEL cells (Fig., B). Inhibition of miR-148a-3p by anti-miR (LNA) leads to elevated TULA-2 expression. A luciferase construct with the 3’UTR miR binding sites was generated. A decrease in luciferase activity was observed in assays with co-transfection of the construct and a miR, supporting the binding between miR-148a-3p and miR-25-3p with TULA-2 3’UTR (Fig., C). Finally, we show that knockdown of TULA-2 caused increased Syk-pY (Y323) level in HEL megakaryocytic cell line in response to FcγRIIA crosslinking by IV.3 (mAb against FcγRIIA) + GAM (Goat anti-mouse F(ab)’2)(Fig., D). These results for the first time show that TULA-2 serves as a negative regulator of the FcγRIIA pathway and that miR-25 and miR-148a may contribute to HIT susceptibility. Studies are in progress for the translation of these findings in vivo.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.