Abstract
Background: This retrospective analysis is focused on the efficacy and safety of radioimmunotherapy (RIT) with Zevalin® in nine patients with recurrent follicular lymphoma (FL) who were treated in a consolidation setting after having achieved complete remission or partial remission with FCR.
Methods: The median age was 63 years (range 46-77), all patients were relapsed with histologically confirmed CD20-positive (grade 1 or 2) FL, at relapse they received FCR every 28 days: F (25mg/m2x 3 days), C (1gr/m2 day 1) and R (375mg/m2 day 4) for 4 cycles. Who achieved at least a partial remission, with<25% bone marrow involvement, was treated with 90Yttrium Ibritumomab Tiuxetan 11.1 or 14.8 MBq/Kg up to a maximum dose 1184 MBq, at 3 months after the completion of FCR. The patients underwent a further restaging at 12 weeks after 90Y-RIT with total body CT scan, FDG-PET/CT and bilateral bone marrow biopsy.
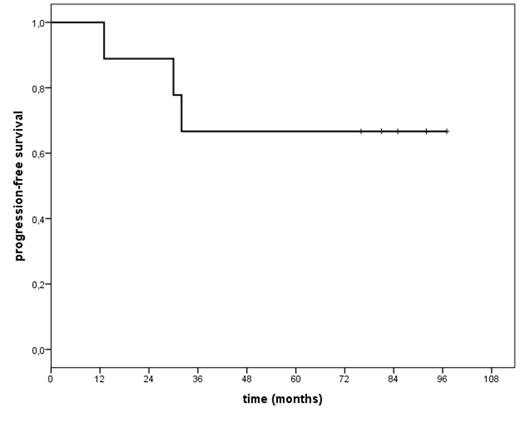
Results: Nine patients have completed the treatment: FCR followed by 90Y-RIT (6 patients at 14.8 MBq/Kg, 3 patients at 11.1 MBq/Kg). After FCR 7 patients obtained CR and 2 PR; after 90Y-RIT two patients in PR converted to CR 12 weeks later. With median follow up of 86 months (range 20-107) since FCR and 79 months (range13-97) since RIT, 3 deaths were observed not related to lymphoma: one for acute renal failure, one for ictus cerebri and one for sepsis; all three deceased patients obtained CR before 90Y-RIT and died still in CR. The median overall survival (OS) and progression free survival (PFS) have not been reached , current analysis has shown that either OS or PFS are 67% at 7 year. The most common grade 3 or 4 adverse events were hematologic, one patient developed herpes zoster infection after 8 months following valacyclovir discontinuation; another patient developed fungal infection.
Conclusions: These results confirm the long term efficacy and safety of 4 cycles of FCR followed by consolidation with 90Y-RIT in patients relapsed with grades 1 and 2 FL and suggest that this regimen could be an option to be used for treatment in this setting of patients, specially at age of 60-75 with no unexpected toxicities.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.