Abstract
Background: BiRd (clarithromycin, lenalidomide, dexamethasone) combination therapy yields >90% overall response rates in newly diagnosed patients with symptomatic multiple myeloma (MM). Long term follow up of the phase II BiRd study revealed 49% of patients went on to receive autologous hematopoietic stem cell transplants (ASCT), either as consolidation or salvage. Here we evaluate progression free survival 2 (PFS2) and overall survival (OS) in our cohort of patients, and assess the impact of ASCT.
Methods: Seventy-two patients with newly diagnosed MM were enrolled on the BiRd trial between Dec 2004 and Nov 2006. BiRd is lenalidomide 25mg PO daily (d1-21), clarithromycin 500mg PO BID, dexamethasone 40mg PO weekly, for 28 day cycle. Patients remained on BiRd until progression of disease, consolidation with autologous stem cell transplant (ASCT), or unacceptable toxicity, and all continued to be monitored through subsequent lines of therapy. We performed a retrospective chart review of all patients enrolled in the BiRd trial. PFS2 was defined as the time elapsed from start of BiRd until progression of disease on 2nd line therapy.
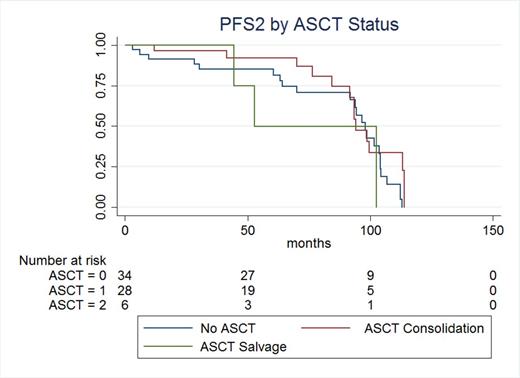
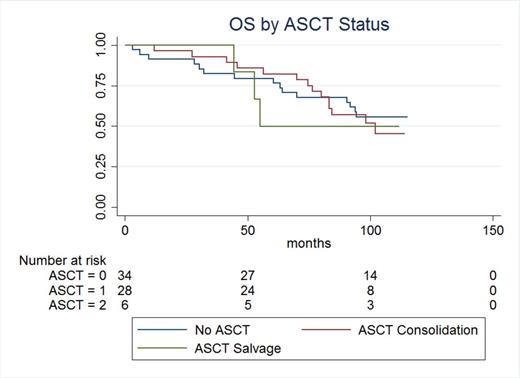
Results: With over 8 years of follow up, 6 patients remain on continuous BiRd, 28 received ASCT consolidation, and another 6 received ASCT as salvage. Nine patients died on induction therapy and the remaining patients received second line chemotherapy. Patients were stratified by no ASCT, consolidation ASCT and salvage ASCT. Median PFS2 was 98 months, 94 months, and 53 months, respectively (p<0.22). Twenty-three patients died during second line therapy. Median OS was not reached, 102 months, and 55 months respectively (p<0.92).
Discussion: Advances in the treatment of patients with multiple myeloma over the past decade have introduced increasing numbers of therapeutic options, improving survival considerably. While multiple myeloma is still considered an incurable disease, patients today will likely receive several lines of therapy. The contribution and timing of each option must be considered. In our cohort of patients, receiving ASCT in the salvage setting trended toward shorter PFS, however failed to reach statistical significance. Consolidation with ASCT in our patient cohort did not translate into survival advantage. The optimal timing and utility of ASCT in the era of novel agents and increasing treatment options warrants further review, and is an area of active investigation.
Rossi:celgene: Speakers Bureau; millenium: Speakers Bureau. Mark:Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Millennium: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Onyx: Research Funding, Speakers Bureau. Pekle:Celgene: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Millennium: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau. Coleman:Celgene: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Millennium: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Onyx: Speakers Bureau. Niesvizky:Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Millennium: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Onyx: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.