Abstract
Introduction: Aged hematopoietic stem cells (HSCs) are known to functionally decline and are prone to development of myeloid malignancies. Recent work has highlighted the twin roles of replication stress and decreased ribosome biogenesis as drivers for the accumulation of DNA damage and senescence. Certain bone marrow failure syndromes, including Shwachman-Diamond syndrome (SDS), Diamond-Blackfan anemia (DBA), and the acquired 5q- syndrome, are characterized by defects in ribosome biogenesis. Furthermore, recent work has suggested a role for p53 activation, through the 5S ribonucleoprotein particle (RNP), in driving cells to senescence following perturbation of ribosome biogenesis.
Methods and Results: Here, we have used multiplexing flow cytometry protocols to define, enumerate, and characterize hematopoietic cells of distinct differentiation stages and lineages in 2 DBA cord bloods and 4 adult bone marrows (2 SDS, 1 DBA, and 1 patient with a diminutive somatic deletion of 5q: ages 27, 32, 40, and 30, respectively), as compared with 4 normal cord bloods and 6 normal adult bone marrows. We included a patient with bona fide MDS (diminutive somatic deletion of 5q including RPS14 in a young adult) to compare with the SDS and DBA patients, who do not meet criteria for MDS.
Our preliminary results revealed significant defects in the primitive HSC and multipotent progenitor (MPP) compartments in both DBA and SDS. Specifically, we found in DBA and SDS bone marrow and cord blood samples (compared to normal controls): significantly decreased numbers of primitive HSCs (Lin-CD34+CD133+CD38-CD45RA-CD49f+CD90+) and MPPs (Lin-CD34+CD133+CD38-CD45RA-CD49f-CD90-); increased levels of apoptosis and dysregulated proliferation; and G0-1/S cell cycle arrest.
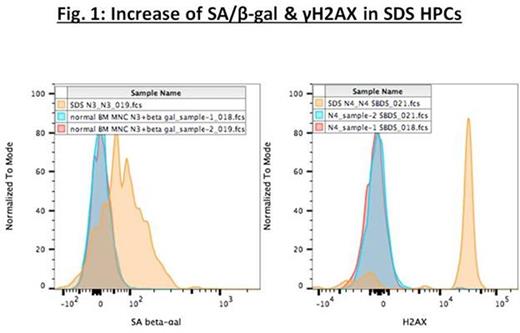
We also found significant increases in senescence-associated β-galactosidase staining and G0-1/S cell cycle arrest in Lin-CD34+ and Lin-CD34+CD38-CD133+ subpopulations in all 4 adult patient bone marrows, as compared with normal adult bone marrows processed in identical fashion [see Fig. 1 for representative data from Lin-CD34+CD133+ hematopoietic progenitor cells (HPCs) from one SDS patient]. Foci of the phosphorylated form of the variant histone H2AX (γH2AX) mark DNA damage, and γH2AX staining was similarly increased in comparison to controls (Fig. 1). The mechanism whereby disturbed ribosome biogenesis induces senescence has been suggested as involving 5S RNP-mediated p53 activation. However, our experiments did not demonstrate increased levels of p53 in the SDS patient marrows, as assessed by intracellular staining. Levels of p16, a well known marker of senescence, were markedly increased in the SDS patient samples, when compared to controls. Finally, in the 2 DBA cord bloods analyzed, there was increased senescence-associated β-galactosidase staining but to a lesser degree than in the adult bone marrow samples (as might be expected with temporal progression).
Discussion: Taken together, our data suggest that ribosomopathies (which often present in childhood) are disorders of premature senescence. Consequent DNA damage accumulation and decreased repair and compensation may account for the development of MDS and acute myeloid leukemia, disorders seen in young ribosomopathy patients that ordinarily are rare in the general pediatric and young adult population.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.