Abstract
Autologous hematopoietic cell transplant (AHCT) plays an important role in the treatment of B-cell Non-Hodgkin lymphoma (B-NHL), both in the upfront and relapsed/refractory settings. In addition to patient and disease-related factors (age, Charlson comorbidity index, disease status pre-AHCT, IPI score at AHCT and high-dose regimen), composition of the infused cell product (higher dendritic cell content, lymphocyte to monocyte ratio < 1) and immune reconstitution after AHCT have emerged as independent predictors of AHCT outcomes. Early absolute lymphocyte count recovery (ALC > 500 cells/μL by day +15 post AHCT) has previously been reported to correlate with superior progression-free survival (PFS) and overall survival (OS) in B-NHL. We conducted a prospective single institution study to further understand the effect of various immune cell subsets and recovery patterns on AHCT outcomes.
A comprehensive immunophenotyping panel including 35 cell subsets was performed in a cohort of 120 consecutive B-NHL patients [Diffuse large B-cell lymphoma (DLBCL) 54%, Mantle cell lymphoma (MCL) 38%] who received AHCT between 1/2008 and 11/2014, at a median of 28 days before AHCT (N=120), and at day +100 (N=95) post-AHCT. Specifically, the immunophenotyping panel included total numbers of T cells, B cells, NK cells and dendritic cells; T cell subsets included the relative proportions of double positive (CD4+CD8+), double negative (CD4-CD8-), CD4+ and CD8+ naïve, CD4+ and CD8+ central memory, CD4+ and CD8+ effector memory, recent thymic emigrants, and T-regulatory cells; B cell subsets included proportions of naïve and memory cells. B-NHL response to AHCT, recovery of ALC by day +15 post-AHCT, PFS and OS were examined and correlated with various immune subsets.
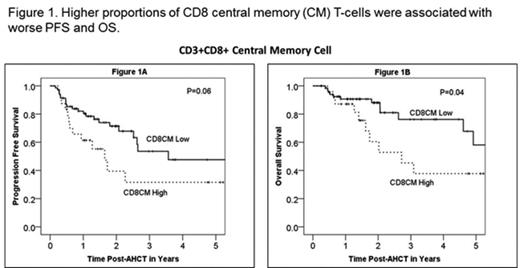
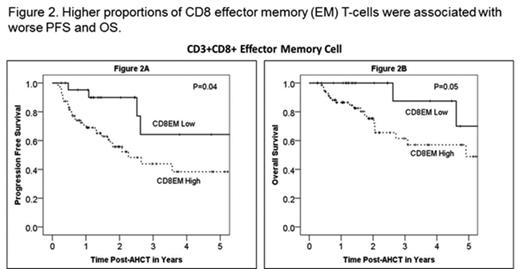
Characteristics in 120 patients pre-AHCT were: median age 59 (range 24-77) years; 71% males; 84% KPS ≥80; disease status pre-AHCT- CR1 43%, ≥CR2 32%, never in CR 13% and relapse ≥1 13%; mobilization regimens: plerixafor+G-CSF 40% and G-CSF alone 60%; high dose regimens: CBV 48% and BuCy 62%. 118 (98%) patients survived beyond day +100 post-AHCT. Day +100 responses: achieved or maintained CR: 83%, less than CR: 3%, disease progression: 15%. Median follow-up was 22.5 (3-42) months. The frequency of different immune subsets either pre-AHCT or at day +100 post-AHCT, PFS or OS were independent of disease histology (DLBCL/MCL) and use of plerixafor as part of mobilization. Unlike an earlier study (Porrata et al, BBMT, 2008), early ALC recovery (by peripheral blood differential count) was not associated with significantly improved PFS or OS in either the whole cohort or individual DLBCL/MCL cohorts. There was no correlation between pre-AHCT immune cell subsets and post-AHCT outcomes. Higher proportions of CD8 central memory (CM) or effector memory (EM) T-cells at day+ 100 post-AHCT were associated with worse PFS and OS (see figures 1 and 2). The frequencies of other immune cell subsets (including NK cells) at day +100 post-AHCT were not associated with attaining CR status post-AHCT, PFS or OS. Further analyses are ongoing to evaluate the association between immune cell subsets in the stem cell product and AHCT outcomes.
This is the first study to examine detailed lymphocyte subsets following AHCT for B-NHL and report a correlation between higher levels of CD8 EM/CM T-cells and decreased PFS and OS after AHCT. Whether relative elevations in CD8 memory T cells directly contribute to disease progression after AHCT for B-NHL or are reflective of an underlying process related to disease progression is uncertain. However, our data suggest that frequency of CD8 memory cells could serve as a biomarker for high risk disease after AHCT for B-NHL.
Paiva:Sanofi: Consultancy; Millennium: Consultancy; Janssen: Consultancy; Celgene: Consultancy; Onyx: Consultancy; Binding Site: Consultancy; BD Bioscience: Consultancy; EngMAb AG: Consultancy. McCarthy:Sanofi: Honoraria, Membership on an entity's Board of Directors or advisory committees; Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees; Onyx: Honoraria, Membership on an entity's Board of Directors or advisory committees; Bristol Myers Squibb: Honoraria, Membership on an entity's Board of Directors or advisory committees; The Binding Site: Honoraria, Membership on an entity's Board of Directors or advisory committees; Karyopharm: Honoraria, Membership on an entity's Board of Directors or advisory committees. Hahn:Novartis: Equity Ownership; NIH/NHLBI: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.