Abstract
Introduction: Ibrutinib is an irreversible first-in-class inhibitor of BTK (Bruton tyrosine kinase) approved for the therapy of relapsed/refractory chronic lymphocytic leukemia (R/R CLL). The drug mediates a transient increase in circulating CLL cells together with reduction in spleen and lymph node size, by both cellular mobilization and apoptosis of resident CLL cells (Herman SE, et al. Blood 2014;123:3286-95). These events occur with important patients' inter-variability (Herman SE, et al. Leukemia 2014;28:2188-96), one cluster of patients presents with greater peak lymphocytosis (resolving between 1 to more than 6 months), while another cluster presents with rapid resolution of lymphocytosis and lymph node/spleen size within 2 months.
Upon such dramatic shifts in disease distribution the first 2 months of therapy (and sometimes lasting >6-12 months), the question of phenotypic changes, sensitivity to monoclonal antibodies (MoAbs), and subclonal diversity of circulating cells remains central for further combination studies. In this study, we evaluated changes in CD5, CD19, and CD20 expression in vitro/in vivo, and peripheral blood side population (SP) cells (a fraction highly enriched in chemorefractory cells, Gross E, et al. Leukemia 2010;24:1885-92) upon ibrutinib therapy. We also investigated whether patterns of lymphocytosis may predict for response to rituximab (RTX) or obinutuzumab (GA101).
Methods: R/R CLL patients (n=25) median prior lines=4, range=2-8), PBMCs were collected before ibrutinib initiation and after 1 and 2 months of therapy. PBMC were seeded at 10 x 106 cells/mL in culture medium and treated for 7 days with 10µg/mL control IgG1 (trastuzumab), RTX or obinutuzumab. The specific percentage of remaining B cells in MoAbs-treated samples was calculated as (absolute number in treated samples/absolute number in control samples) x 100. For each condition, absolute number of remaining B cells =total viable cell number (trypan blue exclusion determination) x % of viable CD19+/CD5+ lymphocytes (flow cytometry determination). For statistical analyses, Student's test (paired, two-sided) was used (*p<0.05;**p<0.01;***p<0.001).
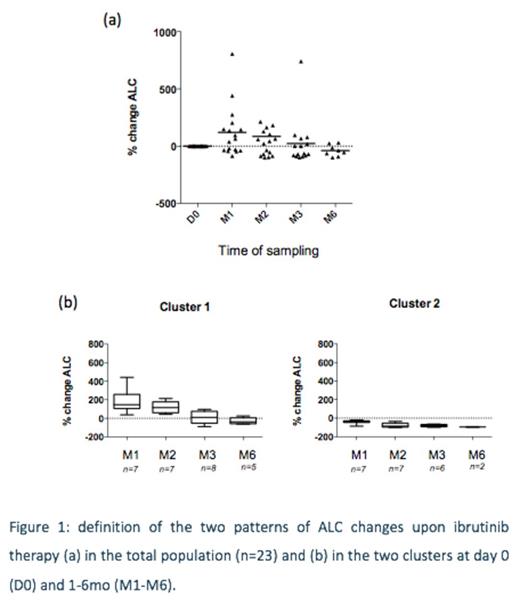
Results: We firstanalyzed patterns ofabsolute lymphocytes count ( ALC) across 23 patients receiving ibrutinib (Fig 1a) to classify them into two clusters as previously published (Fig 1b): Cluster 1 and cluster 2 did not differ significantly in terms of initial lymphocytosis, line of therapy, gender, karyotype, IgHV. Interestingly, the SP fraction in peripheral blood was significantly increased (median: 5/microL before ibrutinib, 10/microL at peak lymphocytosis), suggesting mobilization of resident SP cells, although no apoptosis was detected (in vitro or in vivo) with ibrutinib.
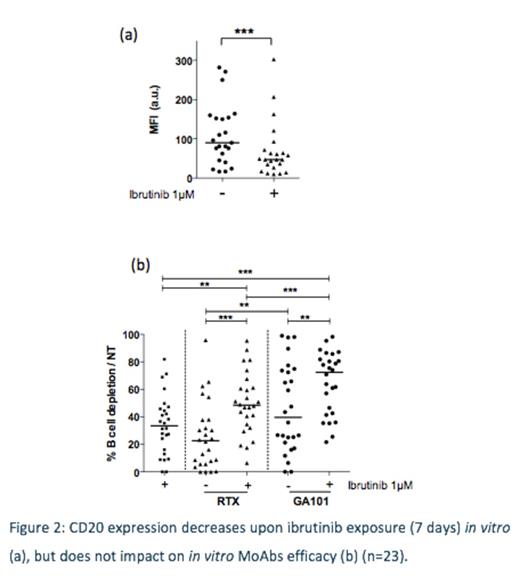
We next assessed CD5, CD19 and CD20 levels in vitro (n=22) and in vivo (n=15) upon ibrutinib therapy. In vitro, ibrutinib significantly reduced CD20 (Fig 2a) and CD19 surface expression, but not CD5; nevertheless anti-CD20 MoAbs still had activity in vitro (Fig 2b). Expression levels were not linked to clusters 1 or 2.
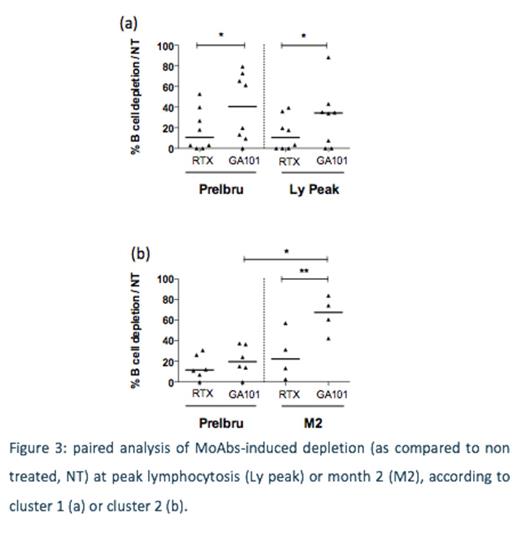
Finally we compared RTX- and obinutuzumab-induced B-cell depletion before administration of ibrutinib, and at various sampling time points (1 to 6 months). Obinutuzumab induced significantly superior depletion at various timepoints than RTX. More interestingly, when analysis was performed from paired samples before/during ibrutinib therapy from the same ibrutinib-exposed patients, only obinutuzumab-induced depletion was increased in cluster 2 (Fig 3).
Conclusions: Ongoing and planned clinical studies evaluate the combination of ibrutinib and obinutuzumab in CLL (first-line and relapsed). Some concerns have emerged due to published preclinical data showing that ibrutinib can interfere with efficacy of therapeutic antibodies. Here, we suggest that ibrutinib-exposed CLL cells, despite wide inter-patient heterogeneity, are targetable with obinutuzumab.
Klein:Roche: Employment.
Author notes
Asterisk with author names denotes non-ASH members.