Abstract
Background
The diagnosis and treatment of CLL may have a great impact on the quality of life (QoL) due to a variety of reasons including disease-related symptoms, infection, effects of therapy and the emotional, socio-economic, and functional effects of living with an 'incurable' illness. The main aim of any treatment is to maximize QoL by inducing remission with minimal short- and long-term toxicity. Balancing disease and symptom control with QoL in elderly patients receiving CLL therapy regimens can be challenging. There is little published QoL data in elderly CLL patients receiving the FCR (fludarabine, cyclophosphamide, rituximab) immunochemotherapy regimen. Our prior study showed no significant difference in health related QoL for first-line therapy of CLL with monotherapy between chlorambucil, fludarabine and cladribine (Mulligan, SP et al. Leuk Lymphoma, 2014).
Aim
We report the QoL assessments of CLL patients enrolled in the ALLG clinical trial CLL5, a randomised, 3-arm, dose de-escalation study of the FCR regimen.
Methods
The treatment schedule has been described with fit elderly patients randomly assigned to one of three treatment regimens: FR5, FCR3, and FCR5 (Mulligan SP et al. iwCLL Abstract, 2015). Treatment was repeated every 28 days for a planned total of 6 cycles. Importantly, an early stopping rule mandating cessation of therapy was included for any patient having grade 3 or 4 toxicity lasting >2 weeks.
QoL assessments using the European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ-C30) were measured at 8 time points: before commencement of therapy, at the end of the 3rd and 6th cycles of treatment, at the final staging and thereafter every 3 months for 12 months.
Results
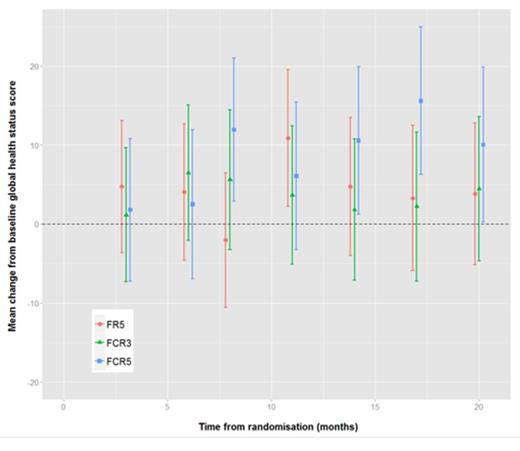
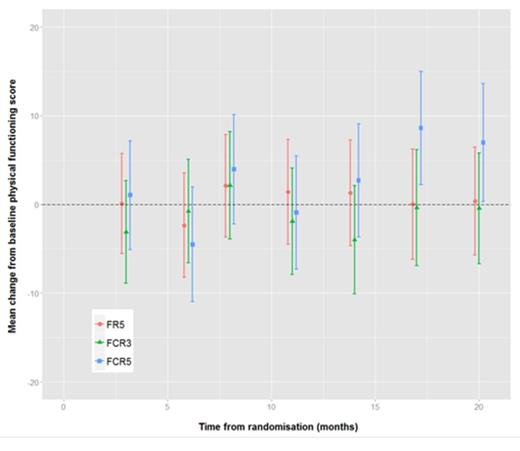
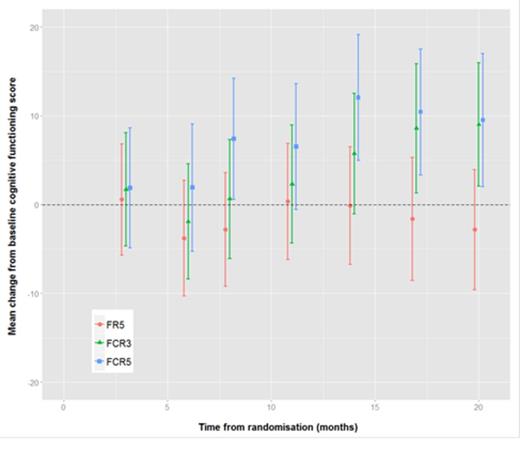
Of the 116 evaluable in the study, 2 patients had no QoL data, 6 patients had only baseline QoL data, and 4 patients had no baseline QoL data, therefore the number of patients included in the QoL analysis was 104. There appeared to be a trend towards improved global health (figure 1), physical functioning (figure 2) and role functioning with full dose (FCR5) towards the end of the follow-up period compared to commencement of therapy. Comparison of cognitive functioning (figure 3) between the three arms showed a statistically significant improvement with FCR5. However, on multivariate analysis there was no statistical significant difference.
Conclusions
Despite a trend favouring full-dose FCR, there were no definitively significant differences in the QoL domains following treatment with FCR-based immunochemotherapy between the three different groups. QoL continues to be a neglected issue, particularly for the typical elderly CLL patient. This study highlights the need for objective QoL assessment as part of all CLL trials, especially those targeted in the typical elderly patient.
Mean change from baseline global health status scores with 95% confidence intervals
Mean change from baseline global health status scores with 95% confidence intervals
Mean change from baseline physical functioning scores with 95% confidence intervals
Mean change from baseline physical functioning scores with 95% confidence intervals
Mean change from baseline cognitive functioning scores with 95% confidence intervals
Mean change from baseline cognitive functioning scores with 95% confidence intervals
Gill:Roche: Research Funding; Sanofi Aventis: Research Funding; AbbVie: Honoraria; Roche: Honoraria. Turner:Roche: Research Funding; Sanofi Aventis: Research Funding. Renwick:Sanofi Aventis: Research Funding; Roche: Research Funding. Latimer:Sanofi Aventis: Research Funding; Roche: Research Funding. Mackinlay:Sanofi Aventis: Research Funding; Roche: Research Funding. Berkahn:Sanofi Aventis: Research Funding; Roche: Research Funding. Simpson:Janssen-Cilag: Honoraria; Celgene: Honoraria; Onyx Pharmaceuticals: Research Funding; Roche: Honoraria. Forsyth:Sanofi Aventis: Research Funding; Roche: Research Funding. Cull:Janssen-Cilag: Membership on an entity's Board of Directors or advisory committees; Amgen: Other: Travel. Harrup:Roche: Research Funding; Sanofi Aventis: Research Funding. Kuss:Sanofi Aventis: Research Funding; Roche: Research Funding. Mulligan:Sanofi Aventis: Research Funding; Janssen: Consultancy, Honoraria, Speakers Bureau; Celgene: Consultancy, Honoraria; Roche: Consultancy, Honoraria, Research Funding, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.