Abstract
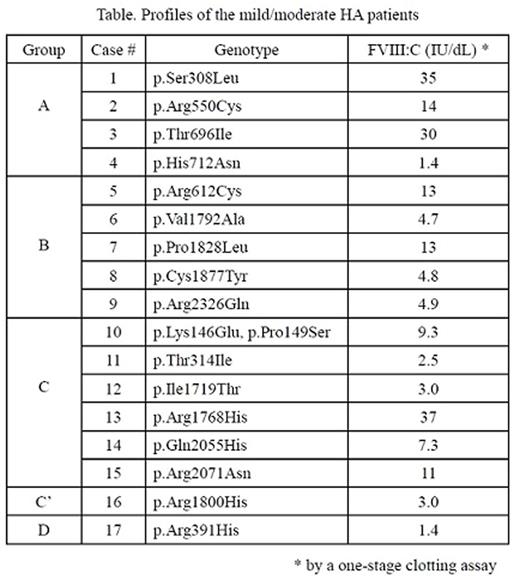
In mild/moderate hemophilia A (MHA), the real hemostatic potential of their innate factor (F) VIII(a) may not be evaluated by FVIII activity (FVIII:C) obtained in a one-stage clotting assay. We often experience the discrepancies between clinical phenotype and FVIII:C in MHA patients. In order to elucidate the hemostatic potentials of the innate FVIII in MHA patients with a simple method, we here have established a plasma-based FXa generation assay (PB-XaG) to clarify the multi-dimensional functions of FVIII(a) in MHA determined by the interaction with the components of tenase complex consisting of FIXa, FX, and phospholipid (PL). XaG was evaluated in the patients' plasmas and in the control plasma dilutions consisting of FVIII-deficient plasma and serial dilutions of recombinant FVIII (1-100 IU/dL) by a chromogenic substrate S-2765 together with human plasma-derived (pd)FIXa (0-20 nM), pdFX (0-200 nM), and PL vesicles (0-114 µM) initiated by thrombin (0.01 nM). The interaction between FVIIIa and each component of tenase complex was evaluated by Michaelis-Menten's kinetic parameters, Vmax and apparent Km (Kmapp). We investigated the PB-XaG in the plasmas obtained from 17 MHA patients, carrying F8 mutations and FVIII:C as shown in the table. The PB-XaG showed that the group A, consisting of the case 1, 2, 3 and 4 with mutations at the residue(s) essential to formation of the inter-domain surface in the FVIII molecule, possessed ~1.8-7.1-fold increases in Kmapp for FIXa (10±0.9, 49±17, 31±6 and 9±3 nM, respectively) compared to FVIII:C-matched control (6.5±1.3 nM), whilst Kmapp for FX and PL in this group were of little difference from that in control, indicative of the reduced affinity for FIXa. The group B, including the case 5, 6, 7, 8 with mutations at the A2 or A3 domain remote from the PL-binding region on the C2 domain (residue 2303-2332) and the case 9 with the mutation at the PL-binding region, demonstrated ~1.7-4.8-fold increases in Kmapp for PL (4.3±2.4, 5.0±1.0, 6.5±1.3, 12±4.0 and 3.0±0.6 µM, respectively) compared to control (2.0±0.4 µM), whilst Kmapp for FIXa and FX in this group were similar to that in control, indicative of the reduced affinity to PL. Noteworthy, the apparent catalytic efficiencies defined as Vmax/Kmapp were 0.033 min-1 for the case 6 and 0.019 min-1 for the case 8 with mutations at the A3 domain, 0.3 or 0.1-fold lower than that for the case 9 (0.19 min-1) with mutation at the PL-binding region. The PB-XaG showed that the group C consisting of the case 10, 11, 12, 13, 14 and 15 demonstrated ~1.6-7.5-fold increases in Kmapp for FX (22±9, 23±9, 24±9, 10±4, 4.6±1.6 and 5.8±3.0 nM, respectively) compared to control (4.5±0.8 nM), whilst Kmapp for FIXa and PL in this group were similar to that in control, indicative of the reduced affinity to FX. Interestingly, the case 16 classified in the group C' showed enhancing affinity with FX represented by a half decrease of Kmapp for FX compared to FVIII:C-matched control (2.0±0.9 and 4.8±1.5 nM, respectively). By contrast, the case 17 with mutation at the thrombin cleavage site categorized in the group D demonstrated any little XaG initiated by thrombin for all axes of FIXa, FX and PL. Taken together, the function of FVIII(a) in MHA could be evaluated multi-dimensionally and clearly classified into the some types of interaction with FIXa, FX, PL and thrombin by the PB-XaG. Our plasma-based XaG assay would be a useful tool for functional analyses of FVIII(a) to clarify the mechanism(s) of hemostatic heterogeneity in mild/moderate HA.
Nogami:Chugai Pharmaceutical Co., Ltd.: Honoraria, Membership on an entity's Board of Directors or advisory committees, Patents & Royalties, Research Funding; F. Hoffmann-La Roche Ltd.: Honoraria, Membership on an entity's Board of Directors or advisory committees; Sysmex Corporation: Patents & Royalties, Research Funding. Shima:F. Hoffmann-La Roche Ltd.: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Sysmex Corporation: Patents & Royalties, Research Funding; Chugai Pharmaceutical Co., Ltd.: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Patents & Royalties, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.