Abstract
Background: Isatuximab (ISA) is an anti-CD38 mAb with potent anti-myeloma (MM) activity given alone or in combination with lenalidomide (LEN) and dexamethasone in relapsed and refractory MM (RRMM). Carfilzomib (CFL) is a second generation proteasome inhibitor approved for the treatment of RRMM. This is an initial report of safety and efficacy data from an ongoing Phase Ib dose-escalation trial combining ISA and CFL in RRMM. (clinicaltrials.gov: NCT02332850)
Objectives: The primary objective is to determine the maximum tolerated dose (MTD) of ISA with standard dose CFL in RRMM. Secondary objectives include characterization of pharmacokinetics, safety (including immunogenicity) and disease response (overall response rate [ORR]; IMWG criteria).
Methods: ISA is given IV every 2 weeks (Q2W), or weekly (QW) for 4 doses then Q2W in combination with standard CFL (20®27 mg/m2: Day 1, 2, 8, 9, 15, 16, every 28 days). Three ISA dosing levels (DL) have been tested: 10 mg/kg Q2W, 10 mg/kg QW x 4 then Q2W and 20 mg/kg QW x 4 then Q2W using a classic 3+3 dose escalation design based on dose-limiting toxicities. Standard premedications (dex 20 mg IV/PO, acetaminophen 650 mg, diphenhydramine 25-50 mg IV/PO and famotidine 20mg IV) were given prior to each ISA infusion.
Results: Eleven patients have been treated across the 3 DL (DL1=3, DL2=3, DL3=5). Treatment disposition: 4 patients have progressed, 7 remain on therapy. This was a heavily pretreated population with a median of 4.5 prior lines of therapy (range: 2-8). All subjects had received prior LEN, 10/11 had received prior BORT and 9/11 had received prior CFL. 10/11 were refractory (refr) to their previous regimen (4 quad refr and 7/11 refr to CFL). 10 patients are evaluable for response. The ORR is 80%: 2VGPR, 6 PR, and CBR is 90% (DL1: 2PR, 1SD; DL2: 3PR; DL3:2VGPR, 1PR, 1MR).
The median # of cycles given is 6 (range 1-20). No DLT's or severe adverse drug reactions have been observed. No dose reduction for ISA or CFL have been needed. The most frequent occurring non-hematologic (NH) adverse events ( occurring in ≥ 15%) at all DLs are dyspnea (45%), fatigue (45%), nausea (45%), pain (back, chest wall and pelvis; 36%), peripheral neuropathy (36%), HTN (27%), cough (27%), anorexia (27%), GERD (18%), constipation (18%), diarrhea (18%), nasal congestion (18%) and hypokalemia (18%); <5% of NH adverse events were Gr 3/4. Heme toxicity was mild; 9% G3/4 anemia, 9% G3/4 neutropenia, 64% G3/4 lymphopenia. The most frequent serious adverse event was Gr 3 pneumonia (18%). Six patients (54%) had infusion reactions (Gr 1 [n=4] or Gr 2 [n=2]) but all completed therapy.
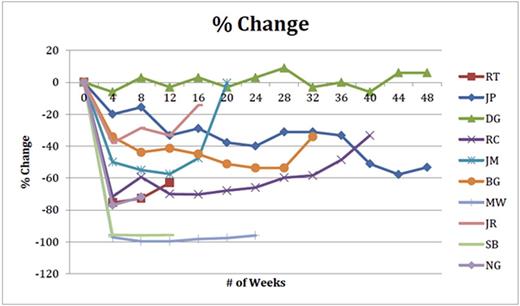
Conclusion: Combining ISA and CFL appears to be well tolerated; no unexpected adverse drug reactions were noted. MTD has not been reached. Encouraging anti-MM activity is reported in heavily pretreated patients with RRMM. Expanded accrual is ongoing, additional safety, pharmacokinetics and response data will be presented. (Figure 1: spider plot of % change in serum m-protein over time)
Martin:Sanofi Aventis: Research Funding; Amgen: Research Funding. Chari:Amgen Inc.: Honoraria, Research Funding; Pharmacyclics: Research Funding; Array Biopharma: Consultancy, Research Funding; Janssen: Consultancy, Research Funding; Takeda: Consultancy, Research Funding; Novartis: Consultancy, Research Funding; Celgene: Consultancy, Research Funding. Campana:Sanofi Aventis: Employment. Hui:Sanofi Aventis: Employment. Wolf:Pharmacyclics: Honoraria; Telomere Diagnostics: Consultancy; Amgen: Honoraria; Takeda: Honoraria; Celgene: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.