Abstract
Introduction: c-Myc is a master transcription factor and one of the most frequently altered genes across a vast array of human cancers including diffuse large B-cell lymphoma (DLBCL), and is thus an attractive therapeutic target . However, no direct inhibitor of c-Myc has been successfully developed for the treatment of any cancer. The c-Myc protein has a short half-life of less than 30 minutes , and the complex secondary structures in the 5' untranslated region (UTR) of MYC mRNA make its translation highly dependent on the eukaryotic translation initiation factor 4F (eIF4F) . eIF4F exists as a complex comprised of the eIF4E, eIF4A, and eIF4G subunits. eIF4E can be sequestered by 4E-BP1, which acts as a "brake" for initiation of mRNA translation . Hyper-phosphorylation of 4E-BP1, caused by upstream signals such as mTORC1, leads to release of eIF4E from 4E-BP1, assembly of the eIF4F complex, and robust mRNA translation. Surprisingly, neither FDA approved mTORC1 inhibitors nor the investigational mTORC1/mTORC2 inhibitor MLN0128 has demonstrated adequate activity in aggressive lymphoma. The therapeutic effects of mTOR inhibition in c-Myc driven aggressive lymphoma remain poorly understood.
Recognizing phosphoinositide 3-kinase (PI3K) and the proteasome pathway are both involved in activating mTOR, we hypothesized that co-targeting the PI3K and proteasome pathways might synergistically inhibit translation of c-Myc. Since both PI3K and proteasome are proven drug targets in blood cancer, such co-targeting strategy may be expeditiously studied in clinical trials for c-Myc driven aggressive lymphoma.
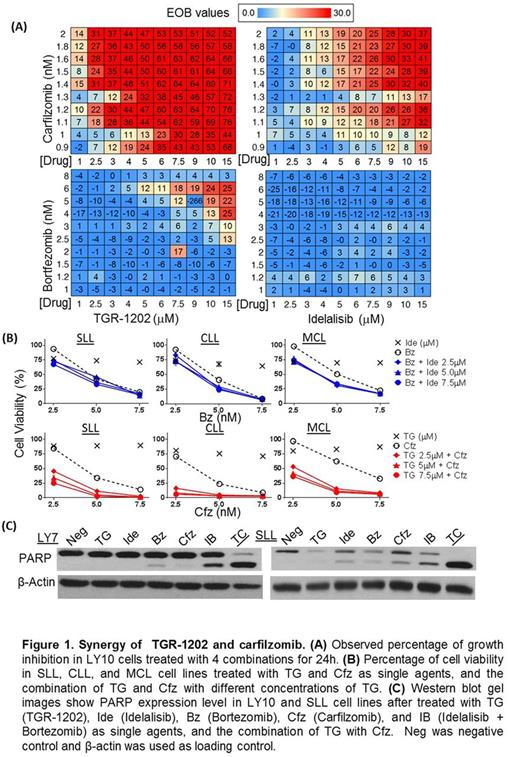
Methods: Cytotoxicity was studied in lymphoma cell lines and primary lymphoma cells using Cell TiterGlo (Promega®). The Bliss additivism model was used to determine the expected inhibition of cell growth and the excess over Bliss (EOB) values. EOB values above 0 indicate synergy, with higher values indicating higher levels of synergy. Expression of c-Myc was investigated at the translation and transcription levels, using a combination of Western blot, qPCR, and a bi-cistronic luciferase reporter we developed to study cap dependent translation. Gene expression profiling (GEP) studies were conducted using RNAseq, and analyzed by the Fisher t-test and running enrichment score (RES) between different treatment groups. Mechanisms of synergy were determined through interrogating the effects of small molecule inhibitors and shRNA targeting regulators of various regulators of 4E-BP1. Structural studies of TGR-1202 were performed by in silico docking, and validated by synthesis of novel analogs of TGR-1202. Activity of TGR-1202 on CK1 epsilon was studied by kinome profiling (Reaction Biology®), cell free kinase assay of CK1e (Promega®), and cell based assay of CK1e autophosphorylation.
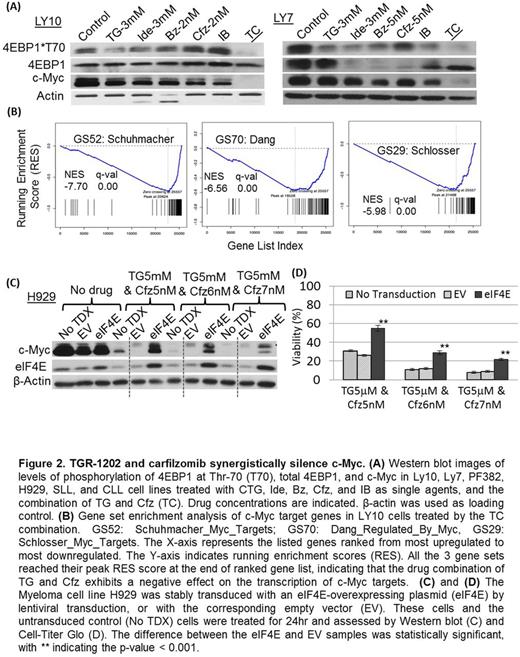
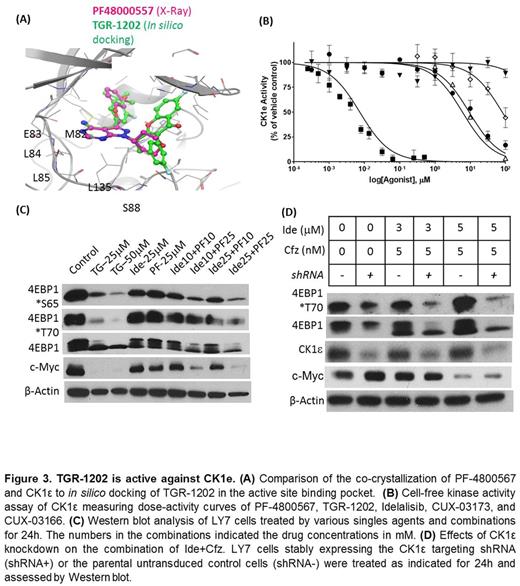
Results: We found that a novel PI3K delta isoform inhibitor TGR-1202, but not the approved PI3Kd inhibitor idelalisib, was highly synergistic with the proteasome inhibitor carfilzomib in lymphoma, leukemia, and myeloma cell lines and primary lymphoma and leukemia cells (Figure 1). TGR-1202 and carfilzomib (TC) synergistically inhibited phosphorylation of eIF4E-binding protein 1 (4E-BP1), leading to suppression of c-Myc translation and silencing of c-Myc dependent transcription (Figure 2). Furthermore, the synergistic cytotoxicity of TC was rescued by overexpression of eIF4E or c-Myc. TGR-1202, but not other PI3Kd inhibitors, was active against casein kinase-1 (CK1) epsilon (Figure 3). Targeting CK1e using a selective chemical inhibitor or shRNA complements the effects of idelalisib, as a single agent or in combination with carfilzomib, in repressing phosphorylation of 4E-BP1 and the protein level of c-Myc.
Conclusion: These results suggest that TGR-1202 is a first-in-class dual PI3Kd/CK1e inhibitor, which may in part explain the preliminary clinical activity of TGR-1202 in aggressive lymphoma not found with idelalisib. Targeting CK1e should become an integral part of therapeutic strategies targeting translation of oncogenes such as c-Myc.
Lentzsch:BMS: Consultancy; Foundation One: Consultancy; Celgene: Consultancy, Honoraria. O'Connor:Seattle Genetics: Research Funding; Mundipharma: Membership on an entity's Board of Directors or advisory committees; Seattle Genetics: Research Funding; Spectrum: Research Funding; Spectrum: Research Funding; Mundipharma: Membership on an entity's Board of Directors or advisory committees; TG Therapeutics: Research Funding; TG Therapeutics: Research Funding; Bristol Myers Squibb: Research Funding; Bristol Myers Squibb: Research Funding; Celgene: Research Funding; Celgene: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.