Abstract
Introduction
Smoldering multiple myeloma (SMM) is an asymptomatic clonal plasma cell disorder. In the last few years several prognostic factors have been shown to identify SMM patients with very high risk of disease progression. Identification of high-risk SMM has assumed greater significance because trials have shown that early therapy can be potentially beneficial to patients. Many risk factors are routinely evaluated in the vast majority of diagnostic centres, such as serum M protein ≥30g/L &/or IgA isotype, immunoparesis, serum involved/uninvolved FLC ratio ≥8 but <100. It has also been shown that replacement of the plasma cell (PC) compartment, defined as ≥95% of total PCs having a neoplastic phenotype by flow cytometry, is a powerful prognostic factor in all plasma cell disorders but there is limited long-term data. The UK's Haematological Malignancy Research Network's (www.HMRN.org) comprises a unified clinical network covering a catchment population of 4 million with a centralised diagnostic facility. Data on newly diagnosed plasma cell disorders has been captured since 2004. The aim of this study is to evaluate the prognostic impact of flow cytometry in newly diagnosed MM.
Methods
All patients newly diagnosed 2004-13 with myeloma (n=2084) in the HMRN's population-based registry were reviewed, and those with baseline flow cytometry analysis that were alive and treatment-free for at least 6 months (n=468) were followed-up until January 2016. Flow cytometry over this time period was consistent with EMN criteria, analysing at least 50k events (typically 200k) using CD138/CD38/CD45 to identify plasma cells and a minimum of CD19/CD56/CD27 to classify as neoplastic or normal phenotype. Biochemistry, immunology and imaging data were performed at the referring hospital while flow cytometry and morphological assessment of aspirate and trephine biopsy were performed centrally. FLC data was infrequently performed before 2008 and therefore follow-up for this parameter is limited. Demographic, prognostic, first-line treatment and outcome data were examined using standard statistical methods.
Results
Of the 468 patients, 30 had bone lesions and one had >60% PC and were excluded from further analysis. 142 cases had >10% PC on the trephine biopsy but <10% PC by aspirate morphology. With a median 6 years follow-up, 13/142 (9.1%) of cases that would have been classified as MGUS without the trephine assessment required treatment during follow-up compared to 100/295 (33.8%) of cases with >10% PC in both aspirate and trephine. In univariate analysis, serum M protein ≥30g/L, IgA isotype, immunoparesis, serum involved/uninvolved FLC ratio ≥8, extent of infiltration on the trephine biopsy, depletion of normal PC (neoplastic PC >95% of total PC) and PC ≥5% of total leucocytes by flow cytometry predicted a significantly poorer treatment-free survival (Log-rank P<0.05). Serum M-protein concentration (HR 2.6, P<0.001), immunoparesis (HR 1.8, P=0.013), trephine involvement (HR 1.6, P=0.001) and PC phenotype/percentage (HR 1.5, P=0.008) retained independent prognostic significance in Cox multivariate analysis (sFLC not included because of insufficient follow-up). The cumulative impact on treatment-free survival for the presence of each of these factors is shown in Figure 1. Age, PC phenotype/percentage and extent of trephine involvement were also predictive of overall survival.
SMM patients with >30g/L M-protein, immunoparesis and PCs >95% neoplastic &/or >5% of leucocytes represented 7% of total SMM patients (29/437) and had a high risk of disease progression with 18/29 (62%) requiring treatment within 2 years of diagnosis.
Conclusions
In a large cohort (>400) of SMM patients with median 6 years follow-up, the results confirm that plasma cell enumeration/immunophenotype is a powerful prognostic factor. Objective evaluation of plasma cell number/phenotype, serum M-protein concentration and immunoparesis identifies high-risk SMM that could be candidates for trials of early therapeutic interventions. Myeloma patients lacking any of the adverse markers have a very low probability of requiring treatment for progressive disease.
Cumulative impact of serum M-protein concentration >30g/L, immunoparesis and PC phenotype/number (PCs >95% neoplastic &/or >5% of leucocytes) on treatment-free survival with one point for the presence of each adverse factor.
Cumulative impact of serum M-protein concentration >30g/L, immunoparesis and PC phenotype/number (PCs >95% neoplastic &/or >5% of leucocytes) on treatment-free survival with one point for the presence of each adverse factor.
Rawstron:AbbVie: Honoraria; BD Biosciences: Other: Remuneration; Janssen: Research Funding; GlaxoSmithKline: Honoraria; Gilead: Consultancy, Honoraria, Research Funding; Genzyme: Honoraria; Roche: Honoraria; Celegene: Honoraria. Owen:Celgene: Consultancy, Honoraria, Research Funding; Takeda: Honoraria, Other: Travel support; Janssen: Consultancy, Other: Travel support.
Author notes
Asterisk with author names denotes non-ASH members.
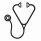
This icon denotes a clinically relevant abstract