Abstract
INTRODUCTION: The antiphospholipid syndrome (APS) has remained an enigmatic condition for over 3 decades. Pathogenic mechanism(s) have not been established, and the disorder has therefore continued to be defined by non-mechanistic assays such as the lupus anticoagulant and antiphospholipid immunoassays. Although evidence from animal models has indicated a role for complement activation in the disease process, it is has been remarkable that activation markers are not consistently detected in human APS plasmas. We hypothesized that this might be due to rapid clearance of activation products and that we might be better able to detect a role for complement in APS through a 2 stage system - i.e. by first allowing APS immune complexes to form on phospholipid vesicles and then measuring whether those vesicles might activate complement in normal serum.
METHODS: Complement activation markers - specifically C5a and the C5b-9 membrane attack complex - were measured in plasmas of 5 groups of plasmas (n= 10 for each group): normal healthy controls, APS with clinical histories for thrombosis (8 with venous thromboembolism (VTE), 1 venous stroke, and 1 arterial stroke), cancer patients without thrombosis, systemic lupus erythematosus (SLE) without APS, and VTE without APS. These were compared to measurements of complement activation induced by phospholipid vesicles that were pre-incubated with the same plasmas. Additional experiments were then done to: 1) characterize whether the effects could be reproduced with purified APS vs control IgGs and β2-glycoprotein I (β2GPI), 2) define the complement activation pathways involved with factor B and C2 deficient sera and with serum in the presence of EGTA and MgCl2, and 3) determine whether the activation could be inhibited by the monoclonal anti-C5 antibody, eculizimab.
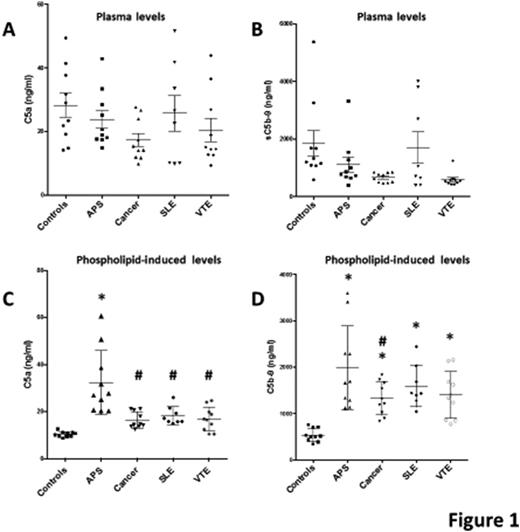
RESULTS: When the plasmas themselves were assayed, we found no significant differences in the plasmas levels of C5a or C5b-9 between APS and controls or among the 5 groups (Fig 1 A, B). In contrast, preincubation of phospholipid vesicles with plasmas resulted in significant elevations of both C5a (Fig 1C) and C5b-9 (Fig 1D) in APS compared to controls. Also, C5a levels in cancer, SLE and VTE were significantly lower than in APS, but above the controls. C5b-9 levels were elevated in APS and the other disease groups compared to controls, with the APS levels that were significantly higher than only the cancer group. The results were confirmed using purified IgGs and β2GPI in place of the whole plasmas. In addition, we found that APS-treated vesicles triggered complement activation via both the classical/lectin pathway and the alternative pathway. Eculizimab completely abrogated the complement activation.
CONCLUSIONS: To our knowledge, this is the first demonstration that APS patient plasmas clearly and consistently activate complement. This was achieved through the novel approach of first incubating the plasmas with phospholipid vesicles which serve as platforms for the assembly of APS plasma-derived protein complexes that, in turn, activate complement. In addition, these findings may provide the basis for a mechanistic biomarker for disease activity in APS, by detecting complement activation at the site of injury.
* P<0.05 vs Controls. # P<0.05 vs APS
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.