Abstract
BACKGROUND: Immune thrombocytopenia (ITP) is an auto-immune disorder characterized by enhanced platelet destruction and, subsequently, increased bleeding risk. It is a chronic condition (cITP) in many patients. In pediatric patients, severe thrombocytopenia may limit activities and those patients who have an insufficient response to therapy may be at risk of severe, potentially life-threatening bleeding complications. Thrombopoietin receptor (TPO-R) agonists have recently emerged as promising therapies for cITP patients who are refractory to other treatments. While eltrombopag (EPAG) is the only TPO-R agonist FDA approved for use in pediatric patients, romiplostin (ROMI) has been used in Phase 3 clinical studies.
OBJECTIVES: This study's objective was to develop an economic model to compare the costs and benefits of EPAG with ROMI in pediatric cITP in the US.
METHODS: A cost-consequence model was developed using a decision tree approach to evaluate the costs of EPAG and ROMI relative to treatment success in previously-treated pediatric patients. Data on platelet count response rate, bleeding events, and adverse events were derived from all relevant identified phase III registered clinical trials; health outcomes were compared via indirect treatment comparison. Costs incorporated in the model included drugs and administration, routine care, rescue medications, bleeding-related adverse events, other adverse events, and mortality costs.
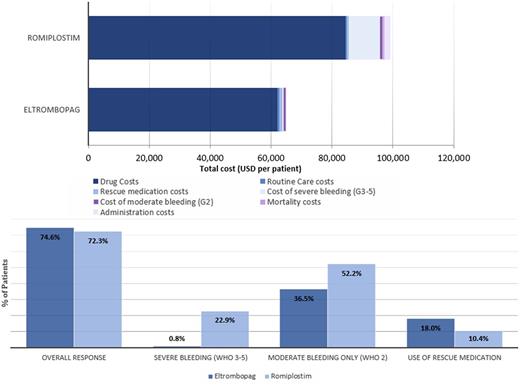
RESULTS: A summary of the cost and efficacy results is presented in Figure 1. The overall estimated cost of EPAG per patient was $66,550, compared to $101,056 for ROMI. EPAG's lower cost compared to ROMI was largely due to lower drug costs ($62,202 vs. $84,396), administration costs ($0 vs. $1,955), and significantly lower costs due to severe bleeding ($354 vs. $10,191). Regarding outcomes, EPAG demonstrated a 22.1% incremental benefit over ROMI when assessing severe bleeding events. Moderate bleeding (WHO grade 2) was also evaluated, where EPAG had a 15.7% incremental benefit over ROMI. EPAG again showed a benefit over ROMI, with an incremental benefit of 2.3% over ROMI when platelet response was assessed.
When considering cost per severe bleeding event avoided, EPAG was dominant over ROMI (less expensive and more effective). EPAG was again dominant over ROMI when assessing the cost per responder and per bleeding event (any grade) . Sensitivity analysis was consistent with the base case findings, with results most sensitive to variations in platelet response rate and changes in primary therapy prices.
CONCLUSIONS: TPO-R agonists are favourable options for the treatment of pediatric patients with cITP who have had an insufficient response to corticosteroids or immunoglobulins. We showed that EPAG was the preferred TPO-R agonist to treat cITP when indirectly compared to ROMI, largely driven by its favorable severe bleeding outcomes and lower drug and administration costs.
Figure 1: Eltrombopag versus Romiplostim, Cost and Efficacy Results
Bhor: Novartis: Employment, Equity Ownership, Research Funding. Roy: Novartis: Employment, Equity Ownership. Elliott: Novartis: Employment. Briggs: Novartis: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.