Abstract
In sub-Saharan Africa, nearly a quarter of a million babies are born with sickle cell disease (SCD) each year. An estimated 50-90% of these babies die before age 5 due to lack of early diagnosis and timely treatment. The World Health Organization estimates that more than 70% of SCD related deaths are preventable with simple, cost-effective interventions, such as early screening followed by affordable and widely available treatment regimens. Here, we present the early clinical testing results of HemeChip, which is the first single-use cartridge-based microchip electrophoresis hemoglobin screening platform. HemeChip was developed by Hemex Health, Inc., based on technology licensed from Case Western Reserve University. HemeChip allows affordable, objective, quantitative screening of hemoglobin variants at the point-of-care. HemeChip works with a drop of finger or heel-prick blood and separates hemoglobin variants on a piece of cellulose acetate paper that is housed in an injection molded plastic cartridge with a precisely controlled electric field.
HemeChip works with a portable reader to produce easily understandable, objective, and quantitative descriptions of the hemoglobin types and percentages present in a blood sample. The HemeChip reader guides the user step-by-step through the test procedure with animated on-screen instructions to minimize user errors. Hemoglobin identification and quantification is automatically done with a custom software on the reader. HemeChip reader records and analyzes the hemoglobin electrophoresis real-time, and it can wirelessly transmit the test results to a central electronic database, if needed.
HemeChip prototype units have been clinically tested and benchmarked against the clinical standard technique in Kano, Nigeria, where the SCD prevalence is the highest in the world. We tested a total of 248 subjects (228 children aged 6 weeks to 5 years in Kano, Nigeria; and 20 adults in Cleveland, Ohio, United States) under institutional review board approval, using both HemeChip and the clinical standard laboratory method, High Performance Liquid Chromatography (HPLC, VARIANT™ II, Bio-Rad Laboratories, Inc., Hercules, California). HemeChip tests were done on eHealth Africa campus in Kano, Nigeria, by trained local healthcare workers using blood samples collected at the nearby Aminu Kano Teaching Hospital. Clinical standard (HPLC) testing was done independently by the International Foundation Against Infectious Disease in Nigeria (IFAIN, Abuja, Nigeria) for the blood samples obtained in Kano or by the University Hospitals Cleveland Medical Center Clinical Laboratories (Cleveland, Ohio) for the blood samples obtained in Cleveland.
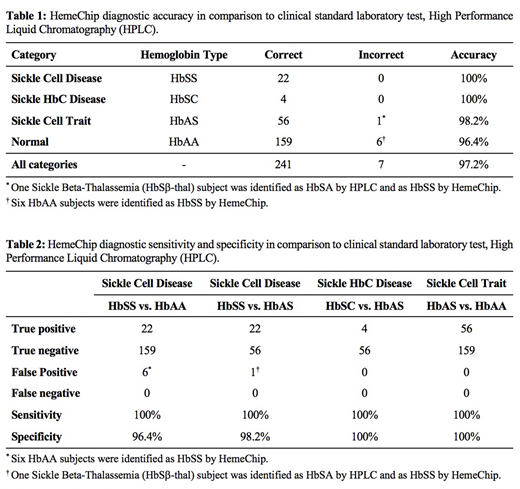
Test results included the following: homozygous SCD (HbSS), heterozygous sickle hemoglobin C disease (HbSC), heterozygous sickle trait (HbAS), and normal (HbAA). HemeChip identified the subjects with HbSS with 100% accuracy, HbSC with 100% accuracy, HbAS with 98.2% accuracy, and HbAA with 96.4% accuracy in comparison to HPLC (Table 1). Overall accuracy of HemeChip was 97.2% in comparison to HPLC for the subjects tested. HemeChip sensitivity was 100% for all hemoglobin variants tested (Table 2), and specificity was 96.4% for HbSS vs. HbAA, 98.2% for HbSS vs. HbAS, 100% for HbSC vs. HbAS, and 100% for HbAS vs. HbAA. Bland-Altman analysis indicated strong agreement between the quantitative HPLC and HemeChip results for hemoglobin percentages, with a mean bias of -3.2%.
HemeChip enables, for the first time, accurate, cost-effective identification and quantification of hemoglobin variants at the point-of-need. HemeChip has been developed based on a versatile, mass-producible microchip electrophoresis platform technology that may address other unmet needs in biology and medicine that require rapid, decentralized hemoglobin or protein analysis, identification, and/or quantification.
Thota:Hemex Health Inc: Employment. Little:PCORI: Research Funding; Hemex: Patents & Royalties: Patent, no honoraria; NHLBI: Research Funding; Doris Duke Charitable Foundations: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.