Abstract
Objective: The Web-Accessible Population Pharmacokinetic Service-Hemophilia (WAPPS-Hemo) platform allows hemophilia treaters to estimate individual PK parameters for clotting factor concentrates using only a few blood samples drawn from their patients. Population pharmacokinetic (PopPK) models used with WAPPS-Hemo are usually built using clinical trial data provided by drug manufacturers. These trials can be restrictive with respect to patient covariates such as age and body weight. On the other hand, real-world pharmacokinetic data submitted to WAPPS captures the breadth of hemophilia patients. We tested the use of post-infusion clotting factor concentrate measurements collected through routine use of WAPPS-Hemo to develop a plasma-derived FVIII (Fanhdi/Alphanate)-specific model for which no trial data was available.
Methods: Plasma factor activity measurements and information on hemophilia A patients were extracted from the WAPPS database. PopPK modeling was completed in NONMEM (ver 7.3, Icon, PLC). Evaluation of model-based Bayesian forecasting to derive the individual PK parameters included 10-fold internal cross validation, limited sampling analysis, and external validation using WAPPS data collected subsequently. The ability of the model to capture age related changes in PK was assessed through separate examination of predictive accuracy for children ≤ 12 yrs and for children and adults >12 yrs of age.
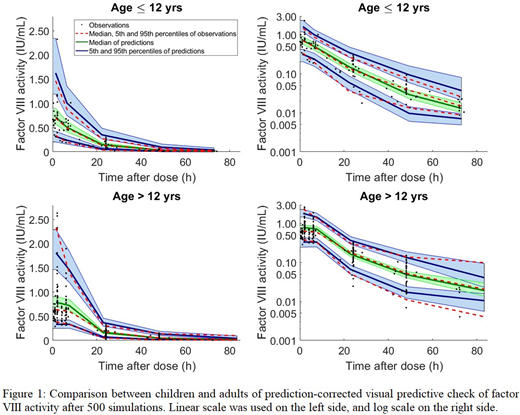
Results: Post-infusion FVIII activity levels from 92 patients were used to derive and internally validate the model. Two-thirds of the patients originated from three centres (Campinas, Brazil; Valencia, Spain; and Santiago, Chile) with the remaining patients from 9 other WAPPS centers. Patients were 1-71 yrs (~33.7% ≤12 yrs) with a body weight range between 9 and 119 kg. Each patient provided between 1-8 activity levels up to 72 hours post infusion (386 data points in total). The final PopPK model followed 2-compartment kinetics with fat-free mass and age as covariates. Clearance was consistent across age until 25 years where clearance declined. The ability of the model to capture the PK in children ≤ 12 yrs was similar in accuracy to patients >12 yrs (Figure 1). Limited sampling analysis demonstrated that sampling strategies using two to three samples with one sample being a 72 hour post-infusion sample produced time-above-2% activity estimates, on average, <5% different than a sampling strategy using 16 post-infusion samples. Bayesian forecasting with additional WAPPS data collected following the model development process (n=10 patients) demonstrated that half-life, clearance, central volume and time-above-2% estimates were within those of the original patient population.
Conclusions: Use of routine clinical care data for model development was feasible and expanded the covariate space (e.g. age) from what is traditionally included in trials. A promising approach would be to supplement clinical trial data with routine clinical data in order to build future PopPK models. On one hand, dense data from clinical trials brings stability to the model and provides a good description of the PK curve and may reduce random error if measured in a central lab; on the other hand, sparse data from routine practice widens the possible observations, inputs and covariates of the model, and could better perform the scope of individual Bayesian forecasting. Our analysis has demonstrated that collecting a few real world samples per patient not only allows accurately determining individual PK parameters, but was also effective for developing a model for a specific brand.
This research was supported by Grifols, a manufacturer of plasma-derived FVIII/VWF concentrates. The WAPPS-Hemo team independently performed the derivation and validation of the model. All authors reviewed and approved the abstract as submitted.
Ozelo:BioMarin: Honoraria, Speakers Bureau; Grifols: Honoraria; Novo Nordisk: Honoraria, Research Funding, Speakers Bureau; Pfizer: Honoraria, Research Funding, Speakers Bureau; Shire: Honoraria, Research Funding, Speakers Bureau; Bioverativ: Honoraria, Research Funding. Iorio:Shire: Other: Alfonso Iorio's Institution has received project based funding via research or service agreements with Shire; Pfizer: Other: Alfonso Iorio's Institution has received project based funding via research or service agreements with Pfizer; NovoNordisk: Other: Alfonso Iorio's Institution has received project based funding via research or service agreements with Novo Nordisk; Grifols: Other: Alfonso Iorio's Institution has received project based funding via research or service agreements with Grifols; Octapharma: Other: Alfonso Iorio's Institution has received project based funding via research or service agreements with Octapharma; CSL: Other: Alfonso Iorio's Institution has received project based funding via research or service agreements with CSL; Bayer: Other: Alfonso Iorio's Institution has received project based funding via research or service agreements with Bayer; Roche: Other: Alfonso Iorio's Institution has received project based funding via research or service agreements with Roche. Spears:Grifols: Employment. Mir:Grifols: Employment. Edginton:Bayer: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.