Abstract
INTRODUCTION: Immune Thrombocytopenia (ITP) is a disease of immune-mediated destruction of platelets and suppression of platelet production. ITP has been historically treated with corticosteroids and/or immune globulins as first-line agents. There are several second-line treatments available, should patients fail to respond to initial treatment or relapse after it is tapered. This study utilizes a national electronic health record (EHR) database to understand clinical outcomes with use of second-line treatments including: thrombopoietin receptor agonists (specifically, eltrombopag and romiplostim), rituximab, and splenectomy.
METHODS: Utilizing the Optum EHR database, we identified patients who initiated a second-line treatment from Jan. 1, 2009 to Sep. 30, 2016 for primary or unspecified ITP. Additionally, patients included in the analysis had the following characteristics: 18 years or older; previous treatment with corticosteroids or immune globulin products; and active in the database for at least 6 months prior to and 12 months post initiation of a second-line treatment. Evaluated outcomes included: (1) platelet counts; (2) bleeding related episodes (BREs); and (3) thrombotic events (TEs), including stroke, transient ischemic attack, myocardial infarction, deep vein thrombosis, and pulmonary embolism. All outcomes were evaluated over the 12-month period following the initiation of second-line treatment. BREs and TEs were defined using ICD codes, collapsed to first three characters, indicating a form of bleeding or thrombotic events. Chi-square test and t-test were used to evaluate differences among the four treatment cohorts (eltrombopag, romiplostim, rituximab, and splenectomy).
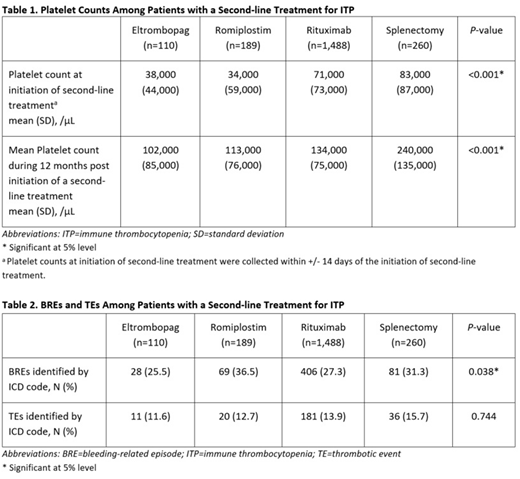
RESULTS: 2,047 patients met the inclusion criteria and used the second-line treatments as follows: eltrombopag, N=110 (5.4%); romiplostim, N=189 (9.2%); rituximab, N=1,488 (72.7%); splenectomy, N=260 (12.7%). The mean age was 60.8 years (standard deviation [SD]: 17.4), with 52.4% female and mean Charlson comorbidity score of 2.1 (SD: 2.1). Compared with platelet counts at the initiation of second-line treatments (collected within +/- 14 days of initiation of second-line treatments), mean platelet counts achieved during the 12 months post initiation of a second-line treatment increased in all treatment cohorts, ranging from an increase of 63,000/μL for rituximab to an increase of 157,000/μL for splenectomy. Mean platelet counts achieved during the 12 months post initiation of a second-line treatment differed significantly across treatment cohorts, ranging from 102,000/μL for eltrombopag to 240,000/μL for splenectomy (p<0.001) (Table 1). The proportion of patients who experienced BREs differed across the treatment cohorts, ranging from 25.5% for eltrombopag to 36.5% for romiplostim (p=0.038) (Table 2). Most common BREs observed included acute posthemorrhagic anemia, GI hemorrhage, hematuria, hemoptysis and hemorrhage, and rectal hemorrhage. TEs were observed in all treatment cohorts ranging from 11.6% for eltrombopag to 15.7% for splenectomy (p=0.744). (Table 2).
CONCLUSIONS: This retrospective real world evidence study compares mean platelet response and burden of both BREs and TEs in ITP patients treated with different second-line treatments. Although there were significant differences in mean platelet counts achieved with the second-line treatments, TEs were observed with similar incidence across all treatments. Patients who received splenectomy had the highest mean platelet counts and also the highest proportion of patients who experienced TEs, though differences in TEs did not reach statistical significance. Incidence proportion of patients with BREs was lower in patients treated with eltrombopag than in patients treated with other second-line treatments. Such differences in outcomes may be useful when selecting treatment in the second-line setting.
Said:Novartis Pharmaceuticals Corporation: Employment. Lal:Optum: Employment. Andrade:Optum: Employment. Nezami:Novartis Pharmaceuticals: Employment. Graves:Novartis: Employment. Roy:Novartis: Employment. Cuker:Kedrion: Membership on an entity's Board of Directors or advisory committees; Synergy: Consultancy; Genzyme: Consultancy; Spark Therapeutics: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.