Abstract
Introduction
Mantle cell lymphoma (MCL) is a heterogeneous disease with a complex genetic landscape. Among genetic anomalies, alterations of several tumor suppressor genes are prognostic markers. The p16INK4A protein, encoded by CDKN2A, is known to bind and inactivate the cyclin-dependent kinase CDK4/6, blocking the phosphorylation of the retinoblastoma protein Rb and inducing cell cycle arrest. The p16INK4A and p53 overexpression are associated with poor prognosis (D. Canioni et al. ASH 2017). Here we compared the expression levels of p16INK4A and p53 in immunohistochemistry (IHC) with the profile (copy number alterations, CNAs) of the genes encoding these proteins, on diagnosis MCL lymph nodes. Results were correlated with patients' outcome in order to identify prognostic biomarkers in MCL.
Methods
All samples (n=86) used in the present work were collected from untreated MCL patients enrolled in the LyMa trial (S. Le Gouill et al. NEJM 2017). IHC was performed for p16INK4A and p53 protein expression assessment on formalin fixed paraffin embedded diagnostic Tissue Micro Arrays. Cut-offs for over expression of p16INK4A and p53 proteins were 10% and 30% respectively (D. Canioni et al. ASH 2016). A pan-genomic copy number analysis was performed with the Oncoscan® SNP-array on DNA extracted from the same samples. Data were compared using chi² tests. Progression free survival (PFS) and overall survival (OS) were studied by log rank test and Kaplan Meier representation.
Results
Patients characteristics (n=86) were similar to the whole LyMa trial population (n= 299) regarding age, gender, Ann Arbor stage and blastoid morphology. Overexpression of p16INK4A was observed in 11% of the patients and was not associated with any deletion of CDKN2A.
There was a significant association between p16INK4A protein overexpression and TP53 mono-allelic deletion (38% vs 7%; p<0.05). CNAs of the CDK4 and RB genes were not associated with p16INK4A protein expression level. Mono and bi-allelic losses of CDKN2A were observed in 19% and 8% of the cases respectively. As expected, bi-allelic loss of CDKN2A (n=7) was associated with a weak p16INK4A expression <5% (p<10-6). However, a similar p16INK4A expression was observed between patients with mono-allelic losses (n=16) and those retaining both copies of CDKN2A (n=65) (p=NS). The 3q26 (BCL6) gains (n=32) were also associated with a higher p16 expression (70% vs 33%; p=0.04). Overexpression of the p53 protein (55% of the patients) was negatively associated with the 15q11 deletion (4% versus 29%; p=0.005) and positively associated with the 1q23 deletion (22% vs 4%; p=0.04) but not with the 17p13 (TP53) deletion.
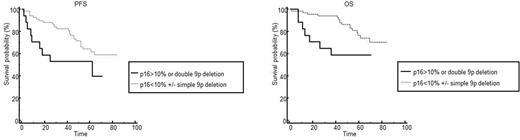
Regarding patients' outcome, early relapse or progression (<1y) were associated with TP53 deletion (HR=5.8; 95%CI 1.0-33.4), CDKN2A deletion (HR=4.0; 95%CI 0.8-22.5), p53 overexpression (HR=7.6; 95%CI 0.9-354) and p16INK4A overexpression (HR=9.0; 95%CI 1.4-56.9) (p<0.05). Overexpression of p16INK4A (p= 0.009) and TP53 deletion (p=0.05) were both found to be associated with a shorter OS in univariate analysis. Only p16INK4A overexpression had an independent impact on OS after multivariate analysis including TP53 and CDKN2A deletion, p16 and p53 expression, (p=0.03; HR=4.3; CI95%: 1.2-15.0). Patients with p16INK4A overexpression or double CDKN2A deletion displayed a worse PFS (p=0.05; HR= 2.7; 95%CI 0.8-8.6) and OS (p=0.02; HR=2.7; 95%CI 0.8-8.6) (Figure).
Conclusion
This work shows that p16INK4A protein expression is correlated with TP53 deletion and BCL6 gain. The p16INK4A overexpression or CDKN2A double deletion could be used as prognostic biomarkers at diagnosis to predict poor response in first line treatment.
Hermine:Hybrigenics: Research Funding; Erythec: Research Funding; Novartis: Research Funding; Celgene Corporation: Research Funding; AB Science: Consultancy, Equity Ownership, Honoraria, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.