Abstract
Introduction: Chronic kidney disease is common in patients with sickle cell disease (SCD) and is associated with increased mortality. As albuminuria and changes in estimated glomerular filtration rate (eGFR) occur well after substantial structural and functional tissue damage have ensued, biomarkers that predict the development of CKD are needed. Metabolite profiling and the bioinformatics tools to study them may aid in the discovery of novel associations and uncover pathophysiological pathways in SCD-related glomerulopathy. In this study, we have performed an exploratory study to identify urinary biomarkers, which may be predictive of kidney damage in children with SCD.
Methods: Consecutive patients with SCD seen at a pediatric chronic care clinic at Kamuzu Central Hospital in Lilongwe, Malawi, between January and May 2015 were evaluated. Patient urine samples were analyzed for metabolites using NMR-based methodologies. Spot urine measurements for albumin/creatinine ratio (UACR) were required to ascertain the degree of albuminuria (normal albuminuria [UACR < 30 mg/ g creatinine] or albuminuria [UACR ≥ 30 mg/ g creatinine]). Descriptive statistics (mean and standard deviation for continuous variables, and count and percentage for discrete variables) were calculated for each of the baseline variables. T-test and Fisher's exact test were performed to compare the baseline measurements between those with and without albuminuria for continuous and discrete variables, respectively. Metabolite measurements were Pareto scaled and quantile normalized. To detect differential metabolites between the normal albuminuria and albuminuria groups, Wilcoxon rank sum test was performed.
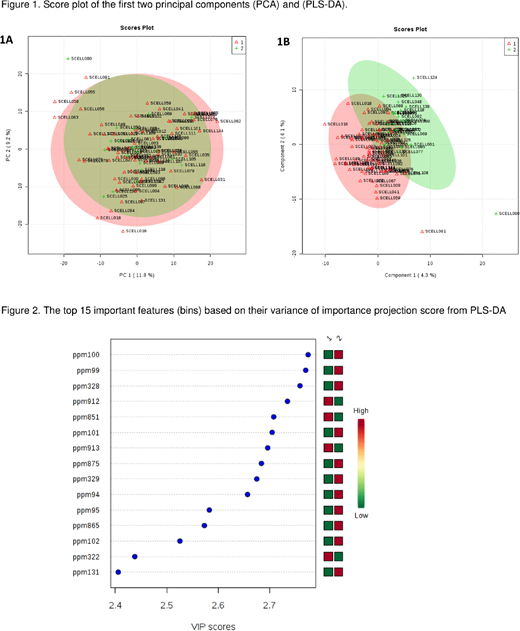
Results: Spot urine samples were obtained in 92 children, 32 (46.4%) of whom were female, average age of 40.6 (±0.98) months, and 23 (25%) patients with normal albuminuria. The patients in both categories were generally similar (age, sex, WBC count, hemoglobin, bilirubin and serum creatinine), except for higher baseline levels of lactate dehydrogenase (p < 0.05) in patients in the albuminuria category. From 1078 bins, we deleted those with low measurements across the majority (>95%) of samples and applied the differential metabolite analysis to 723 bins. Among these bins, 43 metabolites were different in both albuminuria categories (p ≤ 0.05). Although none of the metabolites were significantly different after adjustment for multiple comparisons, the top list is preliminarily identified as ethylmalonic acid, an acidogenic and a metabotoxic branched fatty acid, as an important urinary metabolite differentiating the two groups. High levels of ethylmalonic acid are also found in the urine of patients with short-chain acyl-coenzyme A dehydrogenase deficiency, a fatty acid metabolism disorder. The results remain largely unchanged after important covariates, age, sex, WBC count, hemoglobin, lactate dehydrogenase, bilirubin and creatinine were adjusted. In addition, principal component analysis (PCA) and partial least square discrimination analysis (PLS-DA) were performed (Figures 1A and 1B). Despite the lack of distinct clusters on the score plots, 15 metabolites were identified as "important features" in PLS-DA, based on their variance of importance projection (VIP) score (Figure 2).
Conclusion: Our exploratory study shows that urinary ethylmalonic acid is a potentially important biomarker for the early detection of kidney damage in children with SCD. More studies of larger patient populations with SCD are required to identify urine metabolites which may predict the development of albuminuria.
Key:UniQure BV: Research Funding. Ataga:Global Blood Therapeutics: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Pfizer: Research Funding; Novartis: Consultancy, Honoraria; Modus Therapeutics: Honoraria; Bioverativ: Honoraria, Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.