In this issue of Blood, Streetley et al have used live-cell imaging and high-resolution cryo–electron microscopy tomography to identify CD63+ luminal membrane vesicles in Weibel-Palade bodies (WPBs) in human umbilical vein endothelial cells (HUVECs) and microvascular endothelial cells. In response to an increase in intracellular calcium or cyclic adenosine monophosphate, these membranes are released as so-called exosomes in a fashion similar to that described for platelets and other cells. This is the first report of the presence of intraluminal vesicles (ILVs) in WPBs and the regulated release of exosomes from vasculature endothelial cells.1
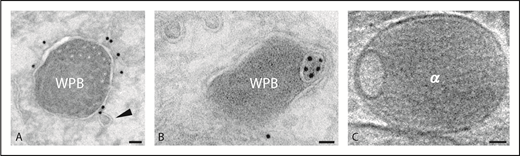
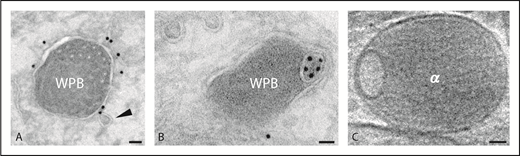
Weibel-Palade bodies and alpha granule. (A-B) Immunogold localization of CD63 on WPBs. Arrowhead indicates a transport vesicle in close position to the WPB (WPB). (C) Tomographic slice of vitrified platelet α-granule (α). Scale bars, 40 nm.
Weibel-Palade bodies and alpha granule. (A-B) Immunogold localization of CD63 on WPBs. Arrowhead indicates a transport vesicle in close position to the WPB (WPB). (C) Tomographic slice of vitrified platelet α-granule (α). Scale bars, 40 nm.
WPBs in endothelial cells belong to the group of lysosome-related organelles (LROs), a heterogeneous group of organelles that share features with lysosomes and secretory granules.2 LROs acquire cargo and membrane components both from the biosynthetic pathway and the endolysosomal system. Formation of WPBs starts at the trans-Golgi network (TGN) and is mainly driven by the assembly of von Willebrand factor (VWF) multimers into tubules that shape the organelle into an elongated structure. Other cargo molecules such as cytokines and the membrane protein P-selectin are included during WPB maturation. Additional components such as the tetraspanin protein CD63 (also called lysosomal-associated membrane protein 3 [LAMP3]) become incorporated at a later stage.3 CD63 shares with LAMP1 and LAMP2 a cytoplasmic Gly-Tyr motif, which serves as a lysosomal-targeting signal.4 CD63 is a well-established component of the late endosomal and lysosomal system in many cells. Although CD63 is found in WPBs, the vast majority of CD63 is present within the complex network of internal membranes characteristic of late endosomes. CD63 in WPBs presumably derives from these endocytic compartments.3 Indeed, as shown by Streetley et al in the present study, extracellular-added fluorescently tagged antibodies directed against CD63 accumulate as discrete microdomains in WPBs. The CD63-enriched microdomains are frequently located at the organelle’s periphery. To further explore the relevance of this finding, the authors used high-resolution cryo-electron tomography of whole-mount HUVECs, vitrified by rapid-plunge freezing. From volume reconstruction analysis, it appeared that these microdomains represent ILVs. The ILVs are topologically separated from the WPB-limiting membrane, and the presence of cytosolic structures such as ribosomes and glycogen therein indicate that the ILVs originate via inward budding of the limiting membrane, a mechanism that has been reported for multivesicular bodies (MVBs). Whether the peripheral location could also represent a prelude to a protrusive-sorting activity at the WPB’s limiting membrane remains to be established. Clathrin-dependent sorting is mainly restricted to the immature TGN-derived WPBs. Together, these findings show that the CD63-rich microdomains in WPBs represent free ILVs, a feature that has also been reported in platelet α-granules.
An important question is how CD63 ends up in the ILVs of WPBs. Membrane fusion with MVBs is a possible mechanism for the formation of WPB-ILVs; MVBs are known to fuse with lysosomes and WPBs have been shown in close apposition to MVBs.5,6 However, the absence of other exosome markers argues against such a mechanism. Although CD63 is enriched on ILVs, it is also found on the limiting membrane of MVBs and WPBs (see figure panels A and B). Steady-state levels of CD63 are present on the plasma membrane and are probably maintained by constitutive trafficking from the TGN and recycling from endosomes. CD63 may be transferred via small transport vesicles formed by endosomal membrane budding.7 The presence of CD63+ transport vesicles in close proximity of mature WPBs as shown in figure panel A (arrowhead) supports such a pathway. These transport vesicles contain AP3, the adapter protein implicated in CD63 delivery to WPBs.7
Platelet α-granules share several properties with endothelial WPBs. Both organelles belong to the group of LROs2 ; are involved in hemostasis, inflammation, and angiogenesis; and share crucial proteins, including VWF and P-selectin. Both organelles also harbor the endo/lysosomal marker CD63. As in WPBs, multimeric VWF in α-granules is assembled in distinct tubular structures and segregated from other molecules at the organelle’s periphery. The tight packing of VWF in long, extended tubular structures is responsible for the tubular shape of WPBs. In contrast, VWF tubules in α-granules are shorter, occur far less frequently, and are not tightly packed. The presence of free ILVs enriched in CD63, lacking other tetraspanins such as CD9 and CD81, is another common feature. As in many other cells, CD63+ ILVs in MVBs are enriched in cholesterol.8 Yet, for unknown reasons, unlike ILVs in α-granules, the ILVs in WPBs appear to be cholesterol-poor.
Activated platelets secrete exosomes through fusion of α-granules, the major storage site of the adhesive glycoproteins VWF and fibrinogen, and MVBs with the plasma membrane.9 The presence of tetraspanin proteins is a common feature of cell-derived exosomes. They have been implicated in adhesive as well as costimulatory and signaling functions. Such a role has been suggested in the modulation of integrin affinity, which could take place on the surface of endothelial cells and activated platelets. In a similar fashion, CD63 may play a role in modulating P-selectin function toward leukocytes.10 Besides common properties such as shared cargo, WPBs and platelet α-granules also have structural differences. Clathrin coats are restricted to TGN-derived early WPBs, but are abundant on mature platelet α-granules. Clathrin on early WPBs and more generally on endosomal compartments reflect an active sorting activity away from the organelle. Mature WPBs lacking clathrin are stable with respect to their content and apparently less dynamic than the α-granules in platelets. Because platelets are known to endocytose plasma proteins, including fibrinogen, an elegant concept could be that clathrin coats on α-granules reflect the organelle’s capacity to recycle αIIb-β3 integrins for constitutive delivery of plasma fibrinogen.
The precise extracellular role of WPB-derived exosomes has yet to be determined. In addition, it will be important to know to what extent WPBs and MVBs contribute to the total secretory exosome pool, and if they have similar extracellular functions. As WPB-derived exosomes have a different lipid and protein composition, it will be interesting to isolate them from the “releasate” of stimulated endothelial cells.
Conflict-of-interest disclosure: The author declares no competing financial interests.