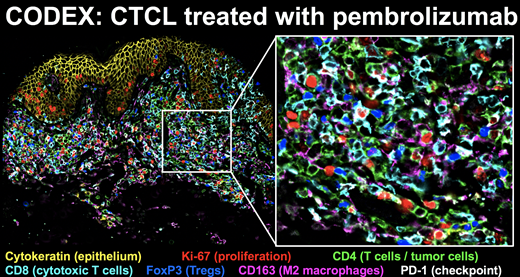
Cutaneous T cell lymphoma (CTCL) is a CD4+ T cell malignancy of the skin with heterogeneous outcomes and limited treatment options. Monoclonal antibodies directed against PD-1, such as pembrolizumab, have shown impressive efficacy in multiple advanced malignancies, and are currently tested in clinical trials in patients with CTCL. Initial data indicate that about half of the patients experience treatment response, whereas the other half are non-responders. Non-responders can be further divided into patients with stable disease versus rapid progressors. It is currently unknown why some CTCL patients respond to pembrolizumab while others rapidly progress, and no predictive biomarkers are available. Single-cell analysis approaches to identify biomarkers of response, for example quantifying the expression of PD-1 on tumor cells vs. reactive immune cells, have not enabled stratification of patients. We therefore hypothesized that more complex spatial cellular interactions within the immune tumor microenvironment (iTME) of CTCL could provide insight into the mechanisms of pembrolizumab response and enable prediction. We applied CODEX (CO-Detection by indEXing) highly multiplexed tissue imaging to study the CTCL iTME in matched biopsies before and after pembrolizumab therapy in 7 responders and 7 non-responders (see the Figure). Using 54 markers simultaneously allowed discriminating malignant CD4+ tumor cells from reactive CD4+ T cells and identified 30 different cell clusters with spatial information, including an M2 macrophage cluster that was enriched in non-responders before therapy. Unexpectedly, in pembrolizumab responders compared to non-responders, PD-1 expression levels were higher in multiple clusters of tumor cells and reactive T cells. Computational spatial analysis revealed ten distinct, conserved cellular neighborhoods in the CTCL iTME that changed in composition and frequency during therapy. Interestingly, one cellular neighborhood to be presented dramatically increased after therapy only in responders. Therefore, highly multiplexed spatial analysis of the CTCL iTME allows discovering novel, predictive biomarkers of immunotherapy response and will pave the way for future studies that functionally address the identified cell types and cellular interactions.
Khodadoust:Corvus Pharmaceuticals: Research Funding. Kim:miRagen: Research Funding; Merck: Research Funding; Medivir: Honoraria, Membership on an entity's Board of Directors or advisory committees; Innate Pharma: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Soligenix: Research Funding; Forty Seven Inc: Research Funding; Neumedicine: Research Funding; Takeda: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Trillium: Research Funding; Elorac: Research Funding; Galderma: Research Funding; Corvus: Honoraria, Membership on an entity's Board of Directors or advisory committees; Portola Pharmaceuticals: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Seattle Genetics: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Kyowa Hakko Kirin: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Eisai: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Horizon: Research Funding. Nolan:Akoya Biosciences Inc.: Consultancy, Equity Ownership, Patents & Royalties.
Author notes
Asterisk with author names denotes non-ASH members.