Background:
Avatrombopag (AVA) is a novel, oral thrombopoietin receptor agonist (TPO-RA) recently FDA approved for the treatment of chronic immune thrombocytopenia (ITP) in patients not responding to prior therapies. Additionally, AVA is approved for the treatment of thrombocytopenia in chronic liver disease patients undergoing a procedure. Unlike eltrombopag, AVA absorption is not affected by dietary fat or polyvalent cations (e.g., calcium, magnesium, iron, etc.) facilitating ease of use by patients and it also has not been associated with hepatotoxicity. A randomized, controlled, double-blind phase 3 study including 32 ITP patients treated with AVA and 17 treated with placebo (PBO) for 6 months was previously published (Jurczak et al., 2018), demonstrating the superiority of AVA to PBO in median cumulative weeks achieving platelet counts (Plt) ≥50,000/µL (12.4 vs. 0.0 weeks, p<0.0001), and a rapid onset of action (65.6% achieved Plt ≥ 50,000/µL on Day 8 for AVA vs. 0% for placebo (p<0.0001). In addition, AVA had a favorable safety profile with headache, fatigue, petechiae, contusion, and upper respiratory tract infection reported as the most common adverse events. Long-term AVA response rates in ITP have not previously been published.
Methods and Aims:
After the phase 3 core study, patients had the option to continue on to receive AVA in an open-label extension phase. Patients were eligible for the extension phase if they had completed the 6-month core study or discontinued treatment due to lack of efficacy in the core study. We analyzed the response rates for patients utilizing two classes of response: 1) a Plt response was defined as Plt ≥50,000/µL as measured during an on-study patient visit, and 2) a complete Plt response was defined as Plt ≥100,000/µL as measured during an on-study patient visit. A subset of "durable responders" who achieved a Plt response for 6 of the final 8 weeks of the core study (34.4% of AVA patients qualified as durable responders), was also analyzed separately to assess for maintenance of this high response rate in the extension phase.
Results:
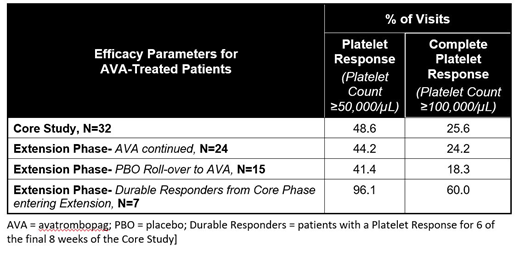
The mean duration of study participation was 23 weeks in the core study and 44 (range, 8-76) weeks overall (core plus extension) including 21 and 19 scheduled visits per patient in core and extension, respectively, for patients completing both studies. In the core study, a Plt response (Plt ≥50,000/µL) was achieved at 48.6% of patient visits for AVA, compared with <0.01% for PBO. In the open-label extension phase for those patients continuing AVA treatment, a Plt response was achieved at 44.2% of visits. For PBO patients who rolled over to AVA in the extension phase, a Plt response was achieved at 41.4% of visits. For all patients ever exposed to AVA (core phase AVA arm and PBO patients rolled over to AVA) during the core study and extension phase, 46.4% of visits achieved a Plt response. In durable responders from the core study (as defined above), a Plt response was achieved at 96.1% of the extension phase visits.
During the core study, a complete Plt response (Plt ≥100,000/µL) was achieved at 25.6% of visits in AVA-treated patients versus <0.01% in the PBO treatment group. During the open label extension phase for patients treated with AVA, a complete Plt response was achieved at 24.2% of visits. For the PBO-treated patients in the core study who rolled over to AVA in the extension phase, a complete response was achieved in 18.3% of the extension phase visits. In total for all patients ever treated with AVA (core phase AVA arm and PBO patients rolled over to AVA), a complete Plt response was achieved at 23.5% of AVA study visits throughout the core study and extension phase. In durable responders from the core study, a complete Plt response was achieved at 60.0% of the extension phase visits. Response rates for AVA-treated patients are summarized in the table below.
Conclusions:
Plt response rates were similar in the core study and extension phase. Durable responders maintained a high rate of Plt response and complete Plt response in the extension phase, providing evidence that AVA-responding patients will continue to respond for extended treatment periods. Additionally, consistent efficacy was noted, as patients in the PBO group who rolled over to active drug demonstrated similar efficacy as AVA-treated patients. AVA-treated patients had substantially higher Plt response and complete Plt response rates than those treated with PBO.
Al-Samkari:Dova: Consultancy, Research Funding; Moderna: Consultancy; Agios: Consultancy, Research Funding. Aggarwal:Dova Pharmaceuticals: Employment. Vredenburg:Dova Pharmaceuticals: Employment, Other: Shareholder. Allen:Dova Pharmaceuticals: Equity Ownership, Other: Chief Medical Officer .
Author notes
Asterisk with author names denotes non-ASH members.