Introduction
Proteasome inhibition has emerged as an important therapeutic strategy in multiple myeloma (MM) and its first-in-class agent, bortezomib, has become a major player in MM treatment. Bortezomib reversibly inhibits the proteasome, inducing an accumulation of ubiquitinated proteins in MM cells, which results in endoplasmic reticulum stress-related apoptosis. However, up to 30% of patients (pts) have been shown to fail bortezomib-based induction and the underlying mechanisms remain poorly understood. As protein degradation by the proteasome, is reliant on protein ubiquitination, uncovering the ubiquitination profile of bone marrow mononuclear cells (BM-MNC) of MM pts, may offer putative biomarkers for predicting the response to treatment and in the future shed new light on disease pathophysiology.
In collaboration with PTM Biosciences, we investigated ubiquitination patterns in BM-MNC samples obtained from MM pts treated with bortezomib-based induction. We hypothesized that ubiquitination patterns at diagnosis would differ between responders vs non-responders and would serve as a platform for identifying potential new therapeutic targets involved in induction of primary resistance.
Methods
BM-MNC samples, collected prior to therapy from 31 NDMM pts, treated with Velcade (bortezomib)-Cyclophosphamide-Dexamethasone (VCD) or VD, were analyzed. Response to treatment was determined after 4-6 cycles of treatment (employing the IMWG criteria), unless progressed earlier.
Functional total cells extract that preserve the cellular enzymatic activity were prepared from each sample and applied to protein microarrays containing ~9000 known proteins (ProtoArray®, Thermo Fisher). Following the exposure of the protein array surface to the sample, enzymes from the sample modified the thousands of proteins spotted on the array, to generate a patient-specific signatures of ubiquitination (Experimental Biology and Medicine 2016; 0: 1-8). The ubiquitination level of every protein was quantified, data was normalized using the quantile normalization method and sorted by descending order for each sample. The ranking position of a protein at each sample within one group was compared to its ranking positions on the other group samples. The proteins differing in their ranking level (p-value ≤ 0.005) between groups were selected.
Results
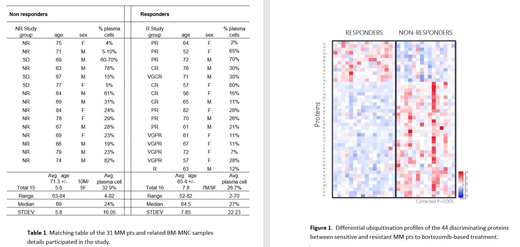
Thirty-one BM-MNC samples obtained from MM pts prior VCD (n=27) or VD (n=4) induction, were tested. The cohort included 16 responding patient that achieved at least PR and 15 non-responding pts that obtained SD or less (Table 1).
PTM analysis initially revealed 241 proteins that were considered as differentially modified between the 2 groups, mostly in the non-responders (153 vs 88). Ubiquitination of a specific protein was determined as "highly discriminating", if shown to be discriminating in at least 50% of the rounds. We identified 44 classifier proteins; 11 that were highly ubiquitinated in the responders and 33 in the non-responders. These 44 proteins were defined as "highly discriminating", providing a unique PTM signature for responders vs non-responders (Figure 1). Among the 44 proteins, 14 (31.8%) were previously suggested to serve as biomarkers in other cancers and 6 other proteins (13.6 %) were found to be related to the ubiquitin/ubiquitin-like or proteasome machinery. At least 3 of these proteins (USP28, SQSTM1 & PSME1) are known to be related to MM and responsiveness to bortezomib treatment. Further validation of 6 of these targets, confirmed their expression in BM-MNC samples in an independent assay. Prospective clinical validation is required to examine the specificity and sensitivity of the identified biomarkers.
Conclusions
Here we provide a proof-of-concept for the ability of ubiquitination profiling of BM-MNC samples to identify putative biomarkers for predicting response to bortezomib-based therapy. Among the proteins identified some were associated with response to bortezomib treatment and other novel candidates require further prospective validation. Algorithm training, validation cohort and investigation of the predictive value of this approach in MM pts treated with other therapeutic regimens are underway.
Our study provides the first example in clinical samples, highlighting the potential significance of this approach in the clinic and offers a novel strategy for proteomics-based personalized medicine.
Tsubery:PTM Biosciences: Employment. Gali:PTM Biosciences: Employment. Gali:PTM Biosciences: Employment. Sela:PTM Biosciences: Consultancy. Merbl:PTM Biosciences: Consultancy, Equity Ownership. Cohen:VBL Therapeutics Â: Employment; Madison Â: Consultancy; Takeda Â: Consultancy; Neopharm Â: Consultancy; Amgen Â: Consultancy, Research Funding; Janssen Â: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.