Background
Acute promyelocytic leukemia (APL) in the elderly has a favourable outcome in a significant proportion of patients (pts). Elderly and frail pts are usually treated with attenuated treatment schedules, mostly "according to medical judgment". The combination of all-trans retinoic (ATRA) and anthracycline-based chemotherapy (CT) has been the mainstay of treatment for many years. Alternative approaches, such as arsenic trioxide (ATO) and gentuzumab ozogamicin (GO) have recently been tested with success in this setting, even though no large series of elderly pts treated with CT-free first-line treatment have been published yet. Moreover, neither a "standard of care approach" nor a specific prognostic score system have been developed to guide physician in choosing the most adequate treatment schedule in this setting.
Objective
Aim of the present preliminary survey was to assess genetic and clinical features of APL in pts over 60 yrs. This cut off reflects the definition of "elderly patient" in most Italian APL GIMEMA trials. Moreover, both the real life outcome and safety data after either conventional anthracycline-based CT or alternative CT-free approaches were analysed. OS, response rate to either regimens and adverse event occurrence were collected.
Methods
This retrospective multicenter REL (Rete Ematologica Lombarda) survey, enrolled a total of 101 consecutive APL pts aged ≥60, treated between 2000-2018. Demographics, clinical data and therapy outcome data were recorded in a dedicated patient's report form. Statistical analysis was performed using the Kaplan Maier method, Log-rank test and Cox regression.
Results
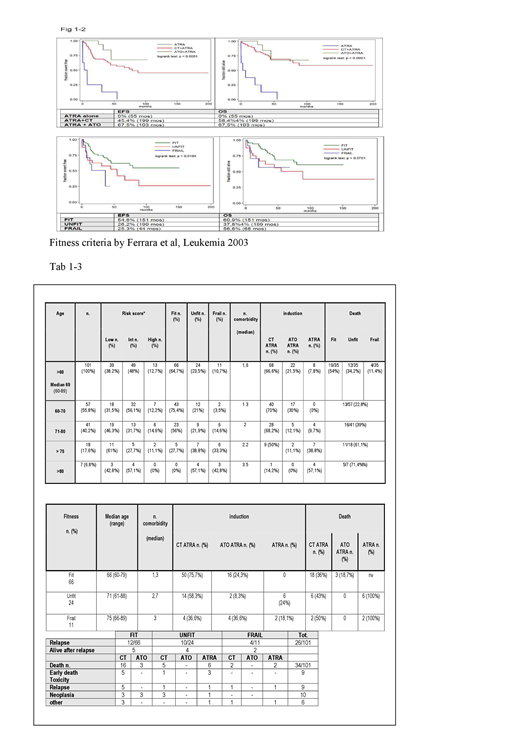
For analysis, pts were grouped into different categories according to age and fitness. Tables 1 and 2 summarise clinical charcteristics and treatment administered. Performance status (PS) and number of comorbidities increase with age. Twentynine of 102 (28,4%) and 9/102 (8,8%) pts had a history of or subsequently developed a solid tumor respectively. High frequency of additive cytogenetic abnormalities was observed as well. CT+ATRA was preferentially administered, mostly with attenuated intensity, to younger pts with better PS, while ATO+ATRA was preferred in pts with reduced cardiac function and ATRA monotherapy was reserved to frail or over 80 yrs old pts. Rates of differentiation syndrome or infections or cardiac events were similar in the different treatment groups. Overall, complete remission (CR) rate after induction therapy was 94.16% [CI95%: 87,5%-97,8% ]. At a median follow-up of 40 mos (range 0-199), the overall relapse rate was 24%. Median time to relapse was 7 mos in 1/8 (12,5%) ATRA monotherapy treated pts and 13 mos (range 5-66) in the 23/68 (33,8%) pts receiving CT+ATRA. No relapse occurred among the 22 pts treated with ATO+ATRA (p 0.005). OS and EFS were significantly associated with pts' age (HR 9% and 7.6%; p<0.001); 35/101 (34,6%) deaths were observed, due to early deaths/toxicity (9/35; 25,7%), relapse or progressive disease (9/35; 25,7%), concurrent neoplasia (10/35; 28,5%), other cause (6/35; 17,1%). Deaths in ATO+ ATRA group occurred after CR, because of neoplasia. Both EFS (median 34 months , range 0.03-199) and OS were significantly higher in the ATO-based therapy group (p<0.0001). (Fig. 1-2)
Conclusion
This survey confirms that elderly and frail APL pts may benefit from ATO-based regimens. Reduced-dose antracycline-based CT, appears to be less effective, with high rate of relapse in elderly pts. Scoring systems and prospective data are needed in order to better define treatment strategies aiming to further improve outcome in this challenging but growing population.
Rossi:Daiichi-Sankyo: Consultancy; Celgene: Membership on an entity's Board of Directors or advisory committees; Amgen: Membership on an entity's Board of Directors or advisory committees; Gilead: Membership on an entity's Board of Directors or advisory committees; Sanofi: Membership on an entity's Board of Directors or advisory committees; Abbvie: Membership on an entity's Board of Directors or advisory committees; Pfizer: Membership on an entity's Board of Directors or advisory committees; Jazz: Membership on an entity's Board of Directors or advisory committees; Astellas: Membership on an entity's Board of Directors or advisory committees; Novartis: Honoraria; Mundipharma: Honoraria; BMS: Honoraria; Janssen: Membership on an entity's Board of Directors or advisory committees; Roche: Membership on an entity's Board of Directors or advisory committees; Sandoz: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.