Background:
Acute myeloid leukemia (AML) is a devastating hematopoietic malignancy caused by differentiation arrest and suppression of apoptosis of immature myeloid cells. The long-term survival of AML under the established therapies remains poor. Differentiation therapy has been developed to promote the normal process of hematopoietic maturation from self-renewing progenitors to terminally differentiated effector cells. The recent discovery of a novel target, the enzyme dihydroorotate dehydrogenase (DHODH), offers differentiation-promoting therapy for the majority of AML (Sykes, D.B., et al.Cell, 2016). DHODH is the rate-limiting enzyme in the pyrimidine biosynthesis pathway. DHODH inhibition was reported to efficiently relieve the differentiation block caused by HoxA9 overexpression in 70% of AML, making this discovery potentially universally applicable for AML patients with diverse genomic alterations. AG-636 is a novel, potent, selective DHODH inhibitor developed by Agios Pharmaceuticals. This small molecule inhibitor has favorable pharmacokinetic properties and is in dose-finding Phase I clinical trials in lymphoma patients (NCT03834584) and is ready to enter a Phase I study in acute leukemia and myeloid dysplasia syndrome. Here, we investigated single agent activity of AG-636 in pre-clinical AML models.
Results:
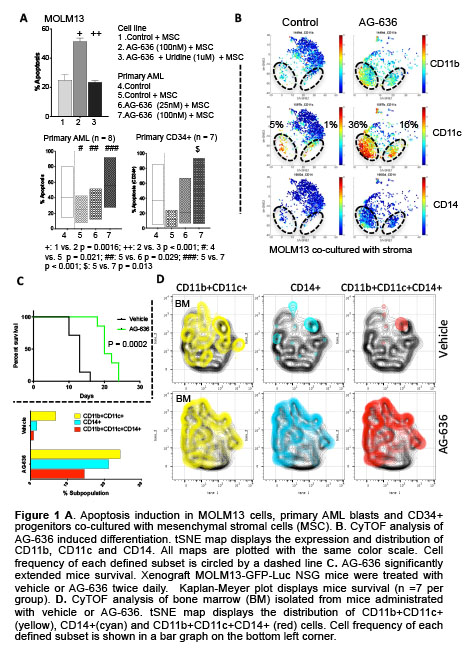
AG-636 inhibited cell proliferation, induced apoptosis in AML cell lines, primary blasts and CD34+ leukemic stem/progenitor cells from AML patients with various genomic alterations cultured under physiologic conditions of stromal support (Fig. 1A). Flow cytometry and multi-parametric mass cytometry (CyTOF) analysis demonstrated that AG-636 reduced bulk AML and facilitated emergence of the differentiated myelo-monocytic cell subset co-expressing CD11b, CD11c and CD14 (Fig. 1B). Both cytotoxic and differentiating effects were rescued by supplementing the DHO downstream metabolite uridine, supporting on-target activity of AG-636 through DHODH inhibition and dependency of AML survival and stemness on the pyrimidine biosynthesis (Fig 1A top).
Mass spectrometric analysis of 166 metabolites confirmed that targeting DHODH by AG-636 resulted in accumulation of the upstream L-dihydroorotic acid and ureidosuccinic acid, and depletion of the downstream metabolites, such as uridine 5'-diphsophate, uridine 5'-monophsophate, CDP and dCMP in pyrimidine biosynthesis pathway. Metabolic profiling further demonstrated the depletion of 5'-phosphoribosyl-N-formylglycinamide in treated cells, indicating the sequential effect of AG-636 on purine biosynthesis and metabolism. Seahorse-based metabolic assay showed inhibition of basal oxygen consumption and ATP generation in AG-636-treated cells, suggesting a contribution of DHODH in coupling of the mitochondria function. Proteomic profiling and immunoblots analysis revealed that AG-636 triggered AMPK activation in response to metabolic stress, and upregulated the expression of TP53, PUMA and NOXA known to regulate mitochondrial integrity. A role for DHODH inhibition in impairment of mitochondria function is of note given the key metabolic dependence of AML cells on OXPHOS/mitochondria function as shown by us and others (Molina J.R. et al. Nat Med, 2018).
In vivo, twice daily administration of AG-636 significantly extended survival in a xenograft MOLM13-GFP-luciferase mouse model (Fig. 1C). Flow cytometry and CyTOF analysis demonstrated that AG-636 induced differentiation of CD11b+CD14+ and CD11b+CD11c+CD14+ monocytes in the bone marrow of treated mice (Fig. 1D). Administration of AG-636 significantly reduced tumor burden, induced differentiation and delayed leukemia progression in two AML patient-derived xenograft mouse models, one harboring mutations EZH2, NRAS and TET3, the other with mutations in ASXL1, BCOR and U2AF1. Daily treatment of AG-636 was well tolerated in all xenograft AML models tested, with a minimal effect on body weight and no significant toxicity recorded over the course of regimen.
Conclusions:
Our preliminary findings demonstrate that AG-636 is highly active against ex-vivo stroma-supported AML and AML stem/progenitor cells and in the in vivo AML xenograft models with diverse genetic subtypes. The significant monotherapy efficacy observed in pre-clinical studies provides the strong rationale for a clinical evaluation of AG-636 in myeloid malignancies.
Kuruvilla:The University of Texas M.D.Anderson Cancer Center: Employment. Kantarjian:Daiichi-Sankyo: Research Funding; Agios: Honoraria, Research Funding; Astex: Research Funding; Actinium: Honoraria, Membership on an entity's Board of Directors or advisory committees; Amgen: Honoraria, Research Funding; AbbVie: Honoraria, Research Funding; BMS: Research Funding; Takeda: Honoraria; Ariad: Research Funding; Cyclacel: Research Funding; Pfizer: Honoraria, Research Funding; Immunogen: Research Funding; Jazz Pharma: Research Funding; Novartis: Research Funding. Andreeff:Daiichi Sankyo, Inc.: Consultancy, Patents & Royalties: Patents licensed, royalty bearing, Research Funding; Jazz Pharmaceuticals: Consultancy; Celgene: Consultancy; Amgen: Consultancy; AstaZeneca: Consultancy; 6 Dimensions Capital: Consultancy; Reata: Equity Ownership; Aptose: Equity Ownership; Eutropics: Equity Ownership; Senti Bio: Equity Ownership, Membership on an entity's Board of Directors or advisory committees; Oncoceutics: Equity Ownership; Oncolyze: Equity Ownership; Breast Cancer Research Foundation: Research Funding; CPRIT: Research Funding; NIH/NCI: Research Funding; Center for Drug Research & Development: Membership on an entity's Board of Directors or advisory committees; Cancer UK: Membership on an entity's Board of Directors or advisory committees; NCI-CTEP: Membership on an entity's Board of Directors or advisory committees; German Research Council: Membership on an entity's Board of Directors or advisory committees; Leukemia Lymphoma Society: Membership on an entity's Board of Directors or advisory committees; NCI-RDCRN (Rare Disease Cliln Network): Membership on an entity's Board of Directors or advisory committees; CLL Foundation: Membership on an entity's Board of Directors or advisory committees; BiolineRx: Membership on an entity's Board of Directors or advisory committees. DiNardo:celgene: Consultancy, Honoraria; daiichi sankyo: Honoraria; jazz: Honoraria; medimmune: Honoraria; syros: Honoraria; notable labs: Membership on an entity's Board of Directors or advisory committees; abbvie: Consultancy, Honoraria; agios: Consultancy, Honoraria. Murtie:Agios Pharmaceuticals, Inc.: Employment. Ulanet:Agios: Employment, Equity Ownership. Konopleva:Calithera: Research Funding; Stemline Therapeutics: Consultancy, Honoraria, Research Funding; Forty-Seven: Consultancy, Honoraria; Eli Lilly: Research Funding; AbbVie: Consultancy, Honoraria, Research Funding; Cellectis: Research Funding; Amgen: Consultancy, Honoraria; F. Hoffman La-Roche: Consultancy, Honoraria, Research Funding; Genentech: Honoraria, Research Funding; Ascentage: Research Funding; Kisoji: Consultancy, Honoraria; Reata Pharmaceuticals: Equity Ownership, Patents & Royalties; Ablynx: Research Funding; Astra Zeneca: Research Funding; Agios: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.