Introduction: Diffuse Large B-Cell Lymphoma (DLBCL) is the most frequent subtype of high-grade lymphoma in Latin American patients. The neutrophil-to-lymphocyte ratio (NLR) has shown to be prognostic in patients with DLBCL in Asia, Europe, and the United States. We previously reported that NLR ≥4 was an adverse prognostic factor for overall survival (OS) in DLBCL patients treated with chemoimmunotherapy. We aim to assess the prognostic and predictive role of NLR ≥4 in a learning cohort of DLBCL patients from South America and confirm the findings in a validation cohort of DLBCL patients from Mexico.
Methods: The study period was from January 2002 through January 2018, and included patients with de novo DLBCL treated with standard chemoimmunotherapy with a curative intent. A cohort of patients from South America (Peru, Argentina, Venezuela, Chile and Colombia) was the learning cohort, a cohort of patients from Mexico was the validation cohort. Univariate and multivariate logistic regression analysis and Cox proportional-hazard regression models were fitted for complete response (CR) as well as for OS in the learning cohort and the validation cohort, separately. Survival curves were estimated using the Kaplan-Meier method and compared using the log-rank test.
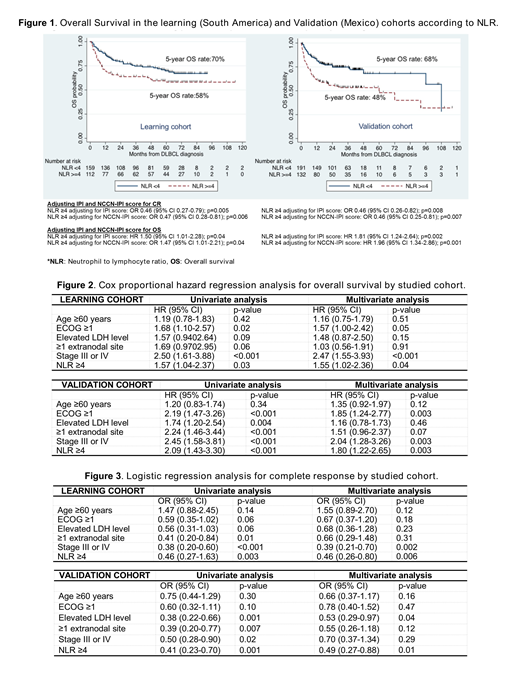
Results: A total of 597 patients with a diagnosis of DLBCL were included. The learning cohort included 274 patients, and the validation cohort included 323 patients.
In the learning cohort, CR rates for DLBCL patients with NLR ≥4 and NLR <4 were 62% and 78%, respectively (p=0.003). In the multivariate analyses for CR, advanced stage and NLR ≥4 were associated with lower odds of CR (OR 0.39, 95% CI 0.21-0.70, p=0.002; and OR 0.46, 95% CI 0.27-1.63, p=0.006, respectively). NLR ≥4 was independently associated with lower rates of CR when adjusted for the IPI and the NCCN-IPI scores (OR 0.46, 95% CI 0.27-0.79, p=0.005; and OR 0.47,95% CI 0.28-0.81, p=0.006, respectively). The 5-year OS rates for DLBCL patients with NLR ≥4 was 58% versus 70% for patients with NLR <4. In the multivariate analyses for OS, advanced stage (HR 2.47, 95% CI 1.55-3.93; p<0.001) and NLR ≥4 (HR 1.55, 95% CI 1.20-2.36; p=0.04) were statistically significant factors associated with worse OS. NLR ≥4 was an adverse prognostic factor after adjusting for the IPI and the NCCN-IPI scores (HR 1.50, 95% CI 1.01-2.28; p=0.04; and HR 1.47, 95% CI 1.01-2.21; p=0.04, respectively).
In the validation cohort, CR rates for DLBCL patients with NLR ≥4 and NLR <4 were 64% and 81%, respectively (p=0.001). In the multivariate analyses for CR, elevated LDH level and NLR ≥4 were associated with lower odds of CR (OR 0.53, 95% CI 0.29-0.97, p=0.04; and OR 0.49, 95% CI 0.27-0.88, p=0.01, respectively). NLR ≥4 was independently associated with lower rates of CR when adjusted for IPI score and the NCCN-IPI score (OR 0.46,95% CI 0.26-0.82, p=0.008; and OR 0.46, 95% CI 0.25-0.81, p=0.007, respectively). The 5-year OS rates for DLBCL patients with NLR ≥4 was 48% versus 68% for patients with an NLR<4. In the multivariate analysis for OS; ECOG ≥1 (HR 1.85, 95% CI 1.24-2.77; p<0.003), advanced stage (HR 2.04, 95% CI 1.28-3.26, p=0.003) and NLR ≥4 (HR 1.80, 95% CI 1.22-2.65; p=0.03) were independent factors associated with worse OS. NLR ≥4 was an adverse prognostic factor after adjusting for IPI score and NCCN-IPI score (HR 1.81, 95% CI 1.24-2.64; p=0.002; and HR 1.96, 95% CI 1.34-2.86; p=0.001, respectively).
Conclusion: This multi-institutional collaborative study identifies and validates an easy-to-use tool, NLR ≥4, as an independent factor predictive of lower rates of CR and prognostic of worse survival, independent of the IPI and the NCCN-IPI scores, in Latin-American patients with DLBCL treated with standard chemoimmunotherapy.
M:Roche-Mexico: Consultancy, Speakers Bureau; Takeda: Consultancy, Speakers Bureau; Merck-Sharp-Dome: Speakers Bureau. Peña:Novartis: Other: Congress inscription and flights; Tecnofarma: Other: Congress inscription and flights; Roche: Other: Congress inscription and flights; Biotoscana: Other: Congress inscription and flights; Janssen: Other: Congress inscription and flights; Pfizer: Membership on an entity's Board of Directors or advisory committees. Rojas:Pfizer: Membership on an entity's Board of Directors or advisory committees; ABBVIE: Membership on an entity's Board of Directors or advisory committees; NOVARTIS: Membership on an entity's Board of Directors or advisory committees; ROCHE: Membership on an entity's Board of Directors or advisory committees. Paredes:Tecnofarma: Honoraria. Ramirez-Ibarguen:Roche-Mexico: Consultancy, Speakers Bureau. Gomez-Almaguer:Amgen: Consultancy, Speakers Bureau; Janssen: Consultancy, Speakers Bureau; Teva: Consultancy, Speakers Bureau; Takeda: Consultancy, Speakers Bureau; Celgene: Consultancy, Speakers Bureau. Castillo:Pharmacyclics: Consultancy, Research Funding; Abbvie: Research Funding; Beigene: Consultancy, Research Funding; TG Therapeutics: Research Funding; Janssen: Consultancy, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.