Dasatinib treatment leads to excellent molecular responses in chronic phase chronic myeloid leukemia (CP-CML). Pleural effusions, an adverse event related dasatinib treatment, may lead to intolerance and drug discontinuations. Strategies aimed at minimising this may improve outcomes. In the Phase III Dasision study, pleural effusion affected ~22% of patients after 4 years of dasatinib treatment at 100mg/d (Cortes et al, 2016 JCO 34(20) 2333-40). The elderly are at particular risk (Latagliata et al 2013 Hem Onc 31(2) 103-9), and there is suggestion that higher dasatinib trough (Cmin) levels may increase the risk of pleural effusions. The randomised OPTIM study (EHA 2014 abstract 5678) has previously reported that patients with Cmin >3nM benefited from dose reductions with preservation of molecular responses. The CML12 (DIRECT) study, run by the Australasian Leukaemia & Lymphoma Group (ALLG) with financial support from BMS, is a single arm phase II study with the aim of minimizing dasatinib related toxicity whilst preserving efficacy using a similar treatment schema to the OPTIM study. Here, we report results of a per-protocol interim analysis based on early molecular response (EMR; BCR-ABL1 ≤10% at 3 months) and MMR (BCR-ABL1 ≤0.1%) at 12 months, both key secondary endpoints of the study. The primary endpoint of the study- the cumulative incidence of pleural effusion at 24 months - is not yet evaluable.
DIRECT initially only enrolled patients >60 years old, predicted to derive the greatest potential benefit from a reduction in toxicity. The protocol was amended after 34 pts were accrued to include patients >18 years old at the recommendation of the trial management committee. All patients started treatment with dasatinib 100mg/day. Dasatinib Cmin was taken at 7, 28 and 56 days after treatment commencement. All Cmin directed dose adjustments were made prior to assessment of BCR-ABL1 at 3 months. Patients sequentially dose reduced to 70mg/day, then to 50mg/day, for Cmin results >3nM. Doses <50mg/day were permitted temporarily only for toxicity management.
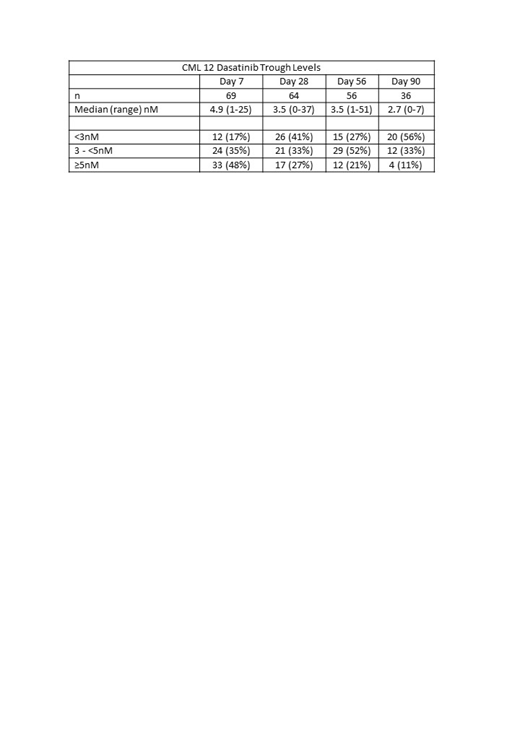
As of June 2019, 71 patients (of 80 planned) from 14 centres have been enrolled, with a median follow up of 7 months (range 0-31). Median age was 64 years (range 21-86) and 48% were female. Sokal risk was low in 40% and high in 9.2%. The median dasatinib Cmin at days 7, 28, 56 and 90 were 4.9, 3.5, 3.5 and 2.7nM respectively (Table). At these time points, 83%, 59%, 73% and 44% of patients had Cmin ≥3nM. There was a trend to lower Cmin after protocol directed dose reduction. The number of patients remaining on 100mg/day after 1, 2 and 3 months of therapy were 11/69 (16%), 5/63 (8%) & 5/55 (9%) respectively.
Molecular response data were available for 48 patients at 3 months and 22 patients at 12 months. At 3 months, BCR-ABL1 ≤ 10% was achieved by 46 of 48 patients (96%), of whom 13 (27%) had achieved MMR (27%). At 12 months, MMR was achieved by 20/22 patients (91%), of whom 7 have achieved MR4.5 (BCR-ABL1 <0.0032%; 32%). The cumulative incidence of MMR by 12 months was 88% (80% CI 75-96%). There was no association between dasatinib dose and molecular response.
Adverse events occurred in 91.5% of patients at all grades - the majority of which were mild. Grade III/IV events occurred in 36.6% and 1.4% of patients respectively. Six of the 71 patients have discontinued dasatinib treatment early, all due to adverse events / dasatinib intolerance. Detailed adverse event data is embargoed until primary endpoint analysis. No patient has progressed to accelerated or blast phase CML.
The DIRECT study demonstrated the feasibility of using dasatinib Cmin levels to optimise dosing. Early molecular response rates are encouraging and predict for excellent achievement of long-term molecular response. Long term efficacy and safety data are awaited.
Yeung:Novartis: Honoraria, Research Funding; BMS: Honoraria, Research Funding; Pfizer: Honoraria; Amgen: Honoraria. Grigg:Abbvie: Membership on an entity's Board of Directors or advisory committees; MSD: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees; Roche: Other: Travel. Shanmuganathan:Novartis: Honoraria, Other: Travel Support; Bristol-Myers Squibb: Honoraria, Other: Travel Support; Amgen: Other: Travel Support; Janssen: Other: Travel Support; Gilead: Other: Travel Support. White:BMS: Honoraria, Research Funding; AMGEN: Honoraria, Speakers Bureau. Branford:Novartis: Consultancy, Honoraria, Research Funding, Speakers Bureau; Bristol-Myers Squibb: Honoraria, Speakers Bureau; Qiagen: Consultancy, Honoraria; Cepheid: Consultancy, Honoraria. Mills:Abbvie: Membership on an entity's Board of Directors or advisory committees; Novartis: Other: Speaker Fees; Amgen: Other: Conference Sponsorship; Specialised Therapeutics: Honoraria; MSD: Membership on an entity's Board of Directors or advisory committees. Shortt:Celgene: Consultancy, Speakers Bureau; BMS: Consultancy, Speakers Bureau; Novartis: Consultancy, Speakers Bureau; Astex: Research Funding; Amgen: Research Funding; Gilead: Speakers Bureau; Takeda: Speakers Bureau. Ross:Celgene: Honoraria, Research Funding; Novartis: Consultancy, Honoraria, Research Funding; BMS: Honoraria, Membership on an entity's Board of Directors or advisory committees. Harrup:Cancer Council of Tasmania: Membership on an entity's Board of Directors or advisory committees; Cooperative Trial Group for NeuroOncolog: Other: Collaborative Clinical Trials Group. Hughes:Novartis, Bristol-Myers Squibb, Celgene: Research Funding; Novartis, Bristol-Myers Squibb: Consultancy, Other: Travel.
Author notes
Asterisk with author names denotes non-ASH members.