Background:
Monocytes and neutrophilic granulocytes are critical for innate immunity, through recognition and phagocytosis of pathogens, and mediation of acute inflammation through secretion of pro-inflammatory cytokines. Furthermore, monocytes constitute an important link between innate and adaptive immunity through antigen presentation to lymphocytes. Patients with chronic lymphocytic leukemia (CLL) have an increased risk of infection, despite often displaying normal neutrophil and monocyte counts. CLL-cells depend on interactions with the immune microenvironment (IME) for proliferation and survival, while inducing changes in surrounding immune cells. However, changes in myeloid cell function in patients with CLL remain sparsely documented. Since the btk-inhibitor ibrutinib impairs B-cell receptor signaling and disrupts CLL-IME interactions (Niemann et al, CCR, 2016), we investigated the impact of ibrutinib and venetoclax on monocyte- and neutrophil phenotype and function in CLL patients.
Methods:
Nine patients treated with ibrutinib 420 mg daily for 8 weeks followed by addition of venetoclax with an initial 5-week ramp up period were included. Blood samples were taken at baseline, after 8 weeks ibrutinib monotherapy, and after another 8 weeks of ibrutinib and venetoclax combination therapy. Immune phenotype was assessed in whole blood by an 8-tube, 10 color flow cytometry panel with custom designed lyophilized antibodies (Duraclone). Immune function was characterized using TruCulture, a whole blood-ligand stimulation assay applying the toll like receptor (TLR) ligands heat killed candida albicans (TLR 2,4,6), lipopolysaccharide (LPS; TLR4), resiquimod (single-stranded RNA-virus analog, TLR7,8), and Poly:IC (double-stranded RNA-virus analog, TLR3), after which the cytokine response was measured. Informed consent from patients and approval from the ethics committee was obtained.
Results:
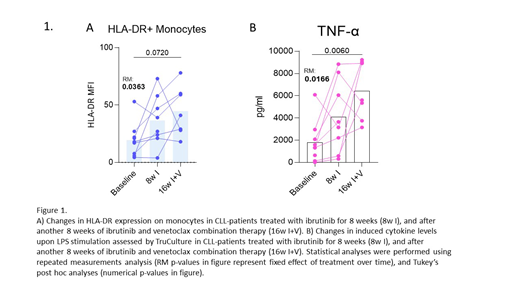
Monocyte and neutrophil counts, as wells as distribution of mature and immature neutrophils, were within normal range at baseline (n=9) and remained unchanged throughout treatment. At baseline, expression of HLA-DR on monocytes was suppressed, but increased significantly upon combination treatment with ibrutinib and venetoclax (p=0.04). HLA-DR expression on neutrophils was high at baseline, remained unchanged upon ibrutinib treatment (n=8), but declined after addition of venetoclax (n=7) (p<0.01). LPS-stimulated TNF-α and IL-6 production was suppressed at baseline, IL-6 levels increased significantly upon ibrutinib monotherapy, and levels of both TNF-α and IL-6 almost normalized upon addition of venetoclax to ibrutinib (p=0.02 for TNF-α, p=0.009 for IL-6).
Conclusion:
Here we show for the first time that myeloid leukocytes in CLL patients exhibit an altered HLA-DR expression at baseline, which is paralleled by an impaired cytokine response to LPS stimulation. Following treatment with ibrutinib and venetoclax, HLA-DR expression increased on monocytes and decreased on neutrophils, both representing normalization. In parallel, LPS-induced TNF-α and IL-6 production improved, likely reflecting an improved monocytic function, as monocytes respond strongly to whole-blood LPS-stimulation. Increased monocyte HLA-DR expression has previously been linked to improved cytokine response (Belge et al, J Immunol, 2002). The contribution of neutrophils to the improved cytokine response is unclear, since the function of HLA-DR expressing neutrophils in CLL is largely unknown. We propose a model where ibrutinib and venetoclax eradicate CLL-cells, disrupt tumor-IME interactions, and maybe directly impact microenvironmental cell signaling, thereby restoring neutrophil and monocyte function and improving immune function. Further investigation of myeloid function in CLL at baseline and upon different treatment regimens is warranted to guide personalized treatment based on modulation of infection-risk.
Svanberg:Novo Nordisk Foundation: Research Funding; Abbvie: Other: Travel grant. Kater:Genentech: Research Funding; Roche: Other: Travel funding, Research Funding; AbbVie: Consultancy, Honoraria, Research Funding. Niemann:Acerta: Consultancy, Research Funding; Roche: Other: Travel grant; Astra Zeneca: Consultancy, Research Funding; Novo Nordisk Foundation: Research Funding; CSL Behring: Consultancy; Janssen: Consultancy, Other: Travel grant, Research Funding; Gilead: Other: Travel grant; Sunesis: Consultancy; Abbvie: Consultancy, Other: Travel grant, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.