Introduction: In the last decade, multiple myeloma (MM) therapy has changed due to management strategies including proteasome inhibitors and immunomodulatory drugs. However, in México these drugs are not affordable for every health care system so there is little data regarding treatment with these agents and their outcomes.
Methods: We retrospectively reviewed the clinical records of patients diagnosed with MM from January 2004 to June 2018, at different hospitals from the Petróleos Mexicanos healthcare services in México. Diagnosis and treatment response were defined according to the International Myeloma Working Group (IMWG) criteria. Baseline and follow-up characteristics and dates of progression, recurrence, death or last follow up were recollected.
Overall survival (OS) was calculated from the date of diagnosis to the date of death from any cause or last follow-up. Event free survival (EFS) was defined from date of diagnosis to the time of progression, relapse or death from any cause or loss of follow-up. Survival curves were estimated using the Kaplan-Meier method and compared using the log-rank test. Risk factors related to EFS and OS were identified through univariate and multivariate analysis by means of Cox proportional Hazard regression model.
Results: A total of 145 patients with MM were included. Median age at diagnosis was 66 years (range 33-94); 38% were female. The most frequent serum monoclonal protein type was IgG. At the time of presentation, 9% of patients had hypercalcemia; 30% renal insufficiency; 83% had anemia < 10 g/dL; 81% osteolytic lesions and 30% had bone marrow plasma cell percentage ≥60%. ISS stages I, II and III were present in 20%, 27% and 53% of cases.
60% of patients received bortezomib-based induction treatment. Only 6% were treated using the combination of bortezomib and lenalidomide plus steroid, and 2% with lenalidomide plus dexamethasone as first line induction regimen. The rest of the cohort received treatment using melphalan, thalidomide or anthracycline-based chemotherapy.
The median follow-up was 47 months (range 2-182). Median OS for the entire cohort was 56 months (95% confidence interval [CI] 42.3-69.7). Six patients (4%) died within six months of diagnosis from renal failure and infections attributed to the disease.
106 patients had evaluable response according to IMWG criteria. Overall response rate was 76% (Complete response 26%; very good partial response (VGPR) 38%; partial response 29%). Only 21 (14%) patients underwent autologous stem-cell transplantation (ASCT). All cases received novel agents as first line regimen and achieved VGPR or better before the procedure. 53 (50%) relapsed during follow up. 38% received second line treatment with lenalidomide-based regimens and 11% were treated with carfilzomib, immunomodulatory drug and steroid combination.
Median OS was 56 months (95% Confidence Interval [CI], 42.3- 69.7). Patients older than 65 years had worse OS (31 months; 95% CI, 19.7-42.3) compared to younger patients (53 months; 95% CI, 25.3-60.7; P= 0.001) as well as those with advanced Durie- Salmon stage or ISS score. Treatment with novel agents had no impact in OS in comparison to other regimens (median 75 months vs 44 months, P = 0.21). On the other hand, in those who received ASCT as consolidation, OS was 130 months compared to 61 months in the group treated with chemotherapy alone. On univariate and multivariate analysis, we identified age > 65 years (hazard ratio [HR] 2.6; 95% IC 1.45- 4.7; P = 0.01) and ASCT (HR 0.3; 95% IC 0.12- 0.77; P = 0.012) as statistically significant factors for survival.
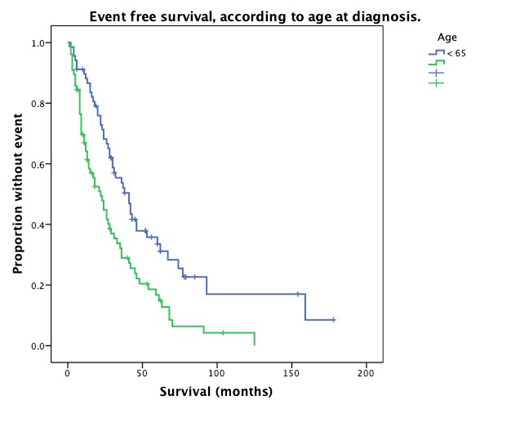
Median EFS was 30 months (95% CI, 20.7- 39.9). We observed a trend towards a better EFS in patients treated using bortezomib and/or lenalidomide-base regimens (36 months; 95% CI, 21.5- 50.5) compared to those who received other treatment strategies (26 months; 95% CI, 13.4-38.6; P = 0.093). Univariate analysis identified age > 65 years (HR 2.1; 95% IC 1.36- 3.42; P = 0.01) as a significant factor for worst EFS; and VGPR or better (HR 0.54; 95% IC 0.45- 0.65; P = 0.001) as well as ASCT (HR 0.34; 95% IC 0.175- 0.67; P = 0.002) as factors associated to better outcomes. Only age and ASCT remained as independent prognostic factors on multivariate analysis.
Conclusion: Outcomes on this study are similar to the reported by other series in México and Latin America. These findings reinforce the need to expand and improve access to better treatments and health coverage in México.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.