Introduction
High-risk subtypes of DLBCL, including high-grade B-cell lymphomas (HGBCL) with rearrangements of c-MYC (MYC-R), BCL2 and/or BCL6 [or double hit and triple hit lymphomas (DHL/THL)], double expressor lymphoma (DEL), and patients with International Prognostic Index (IPI) scores of 3-5 have a poor prognosis with standard R-CHOP chemotherapy. Treatment intensification with DA-EPOCH-R did not improve outcomes when compared to R-CHOP in the CALGB 50303 trial (Bartlett et al JCO 2019). However, high-risk patients were underrepresented as 36% of patients had IPI scores of 3-5, 0.61% of patients had DHL or THL, 2.6% of patients had MYC-R, and 8.6% of patients had DEL (Bartlett et al JCO 2019). Our purpose was to retrospectively compare DA-EPOCH-R to R-CHOP in the treatment of high-risk DLBCL.
Methods
Patients ≥ 18 years old who received either DA-EPOCH-R or R-CHOP at Moffitt Cancer Center between 1/2010 - 9/2019 were included. Patients with a diagnosis of DLBCL with IPI score of 3-5, DHL/THL, and/or DEL were included. Patients with primary mediastinal B-cell lymphoma (PMBCL), DLBCL with testicular or central nervous system (CNS) involvement, grade 3b follicular lymphoma, Richter's transformation or IPI score < 2 were excluded. The primary endpoint was complete response (CR) rate and secondary endpoints included progression-free survival (PFS), overall survival (OS), CNS relapse rate, and adverse events (AE). PFS and OS were estimated using Kaplan-Meier curves and differences were established using the log-rank test. Statistically significant difference was considered if p < 0.05.
Results
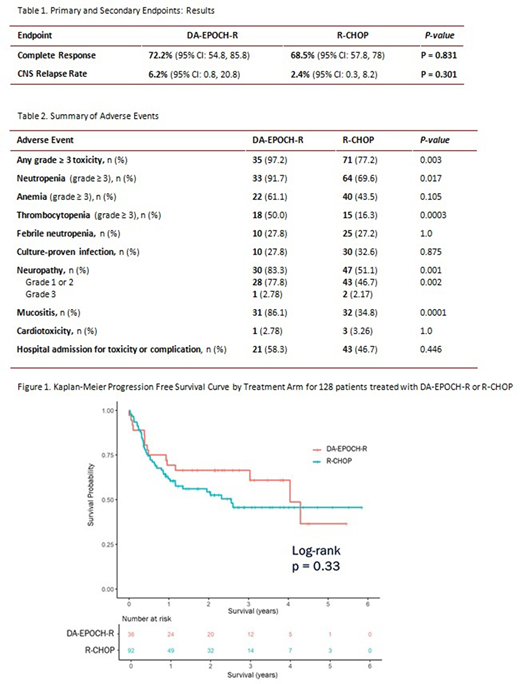
The study included 128 patients: DA-EPOCH-R (n=36) and R-CHOP (n=92). General characteristics were similar between groups and included: median age = 66 years (33-85), stage III/IV = 89%, IPI score 3-5 = 87.6%, non-germinal center B-cell (GCB) = 32.8%, DEL = 26.6%, and DHL/THL = 18%. Of note, more patients in the DA-EPOCH-R group were < 60 years old, had MYC-R, and/or were DHL/THL or HGBCL, NOS cases.
The CR rates for DA-EPOCH-R and R-CHOP were 72.2% (95% CI: 54.8, 85.8) vs. 68.5% (95% CI: 57.8,78.0), respectively (p=0.831). In a pre-planned subgroup analysis, the CR rates across subgroups, including IPI score 3-5, DEL, and DHL/THL, were not different. There was no difference in PFS between DA-EPOCH-R and R-CHOP (p= 0.33; Figure 1). The 1-year (y) PFS for DA-EPOCH-R and R-CHOP was 69% (95% CI: 56.0, 86.0) and 62% (95% CI: 52.0, 73.0), respectively. The 3 y PFS for DA-EPOCH-R and R-CHOP was 66% (95% CI: 53.0, 84.0) and 46% (95% CI: 35.0, 59.0), respectively. PFS was higher in the DA-EPOCH-R arm for patients with MYC-R (p=0.0224) and DHL/THL (p=0.045). There was no difference in OS between DA-EPOCH-R and R-CHOP (p=0.83). The 1 y OS was 77% (95% CI: 65.0, 92.0) for the DA-EPOCH-R group compared with 81% (95% CI: 73.0, 90.0) for the R-CHOP group. The 3 y OS was 71% and 65% for the DA-EPOCH-R and R-CHOP groups, respectively. The CNS relapse rate was 6.2% in the DA-EPOCH-R group and 2.4% in the R-CHOP group (p=0.301). Of the 4 patients with CNS relapse, 3 were DHL, THL or DEL.
There were more grade ≥ 3 AEs in DA-EPOCH-R vs. R-CHOP (97.2% vs. 77.2%; p=0.003). There was significantly more grade ≥ 3 hematologic toxicity in the DA-EPOCH-R group regarding neutropenia (DA-EPOCH-R 91.7% vs. R-CHOP 69.6%; p=0.017) and thrombocytopenia (DA-EPOCH-R 50% vs. R-CHOP 16.3%; p=0.0003). Neuropathy and mucositis were significantly more common in the DA-EPOCH-R arm.
Conclusions
R-CHOP remains standard of care for high-risk DLBCL subgroups when compared to DA-EPOCH-R (such as non-GCB DLBCL, DEL, and patients with IPI score > 3). Patients with DHL/THL or MYC-R had greater benefit from DA-EPOCH-R compared to R-CHOP. DA-EPOCH-R was associated with increased rates of AEs, including significantly more hematologic toxicity.
Chavez:AstraZeneca: Speakers Bureau; Celgene: Consultancy; Bayer: Consultancy; Kite, a Gilead Company: Consultancy, Speakers Bureau; Novartis: Consultancy; Merck: Research Funding; Morphosys: Consultancy, Speakers Bureau; BeiGene: Speakers Bureau; Karyopharm: Consultancy; Genentech: Speakers Bureau; AbbVie: Consultancy; Verastem: Consultancy; Pfizer: Consultancy; Epizyme: Speakers Bureau; Gilead: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.