Introduction: POEMS syndrome (polyneuropathy, organomegaly, endocrinopathy/edema, myeloma protein, skin changes) is a rare disorder associated with plasma cell neoplasia where an overproduction of proinflammatory cytokines such as vascular endothelial growth factor (VEGF) upregulates vasculogenesis and increases vascular permeability resulting in multiple symptoms of the disease e.g. edema and hemangioma. Although the limited-stage disease can be treated with radiotherapy, treatment for the more advanced disease remains unclear. Here we offer a systematic review of the efficacy and safety of treatment regimens used to treat POEMS syndrome in the adult population.
Methods: Databases i.e. PubMed, Embase, Web of Science, and clinicaltrials.gov were searched since inception through May 15th, 2020, following the PRISMA guidelines. Out of 421 studies, 8 articles (5 prospective clinical trials, and 3 retrospective studies) were included. Phase I clinical trials, abstracts, case reports, case series, and review articles were excluded.
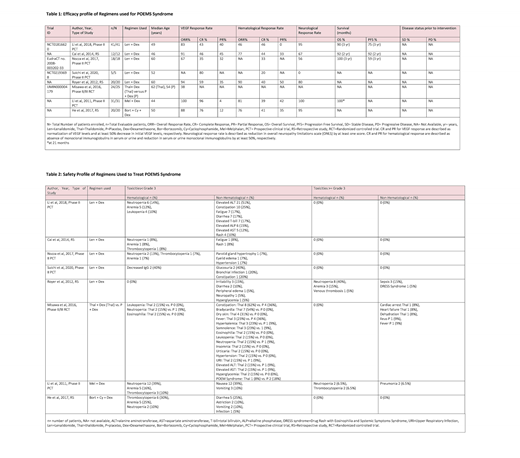
Results: A total of 171 patients (21-79 years of age) were evaluated in 8 clinical studies.
Immunomodulator Based Regimens:
In the 5 clinical studies (N=96) where lenalidomide (Len) plus dexamethasone (Dex) were used, the hematological (heme) complete response (CR) ranged from 20% to 46%. Heme CR was defined as the absence of monoclonal immunoglobulins in serum or urine. VEGF CR defined as normalization of serum VEGF levels ranged from 35% to 80%. Neurological response defined as the improvement in the overall neuropathy limitations scale (ONLS) by at least one score ranged from 0% to 95%. These studies reported 2-3 year progression-free survival (PFS) and overall survival (OS) of 59% to 92% and 90% to 100%, respectively. In the study by Suichi et al. (N=5), VEGF CR was reported to be 80%. However, no neurological improvement was seen in these patients.
Proteasome Inhibitor Based Regimens:
In the study by He et al. (N=20) bortezomib in combination with cyclophosphamide and Dex yielded heme, VEGF, and neurological responses in 76%, 88%, and 95% of the patients, respectively. No grade ≥3 treatment-related adverse effects (TRAE) were seen.
Conventional Chemotherapy Based Regimens:
In the phase II trial conducted by Misawa et al. (N=25) the reduction in serum VEGF levels, as well as clinical response as measured by improvement in muscle strength, was higher in the thalidomide arm as compared to the placebo arm. Grade ≥3 TRAE reported in the treatment arm were cardiac arrest, heart failure, and dehydration in 1 (8%) patient each. In the study by Li et al. (N=31) melphalan (Mel) in combination with Dex achieved heme, VEGF, and neurological response rates of 81%, 100%, and 100%, respectively. Twenty percent of the patients experienced grade ≥3 TRAE.
Conclusion: Immunomodulator and chemotherapy based regimens appear to have a reasonable safety and efficacy profile. Lenalidomide and melphalan based therapies were most effective. Proteasome inhibitors based regimens yielded efficacious results without significant toxicity. More studies with existing and newer combinations with autologous stem cell transplantation are warranted.
Anwer:Incyte, Seattle Genetics, Acetylon Pharmaceuticals, AbbVie Pharma, Astellas Pharma, Celegene, Millennium Pharmaceuticals.: Honoraria, Research Funding, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.