Abstract
Background
Although classic Hodgkin lymphoma (cHL) is highly curable with current treatment paradigms, there is still 20-30% of patients who will experience therapeutic failure (refractory or relapse). Tislelizumab, a monoclonal antibody targeting programmed cell death protein 1, is showing promise in the clinic with obvious response in relapsed/refractory cHL (RRcHL) patients on monotherapy. Further improvements in antitumor efficacy as salvage therapy may require exploration of a tislelizumab-based combination regimen. We conducted a phase II multi-center, open-label, non-randomized study (ChiCTR2000033441) to investigate the efficacy and safety of tislelizumab with gemcitabine plus oxaliplatin (GemOx) in patients with RRcHL.
Methods
Patients who had at least one previous therapy for cHL and were relapsed or refractory were eligible. Enrolled subjects received up to eight courses of gemcitabine (1g/m 2 on day 1) and oxaliplatin (100 mg/m 2 on day 1) combined with tislelizumab (200 mg on day 2) at 21-day intervals. Then, the cohort was divided into a tislelizumab maintenance group (every 2 months for 2 years) and an autologous hematopoietic stem cell transplantation group based on the choice of the investigators. The primary endpoint of this study was the best complete response rate (CRR), and secondary endpoints included overall response rate (ORR), progression-free survival (PFS) at 12 months, and safety profile.
Results
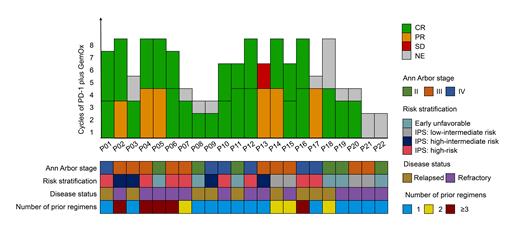
As of July 2021, a total of 22 patients (median age of 33 years old) were included. The predominant histologic subtype was nodular sclerosing cHL. Early-stage cHL was found in six patients and late-stage cHL in 16 (two of them with bulky disease). Correspondingly, 59.1%, 18.2%, and 22.7% of patients had 1, 2, or ≥ 3 prior therapies, respectively, and more than half of patients (59.1%) reported refractory to the latest treatment. The efficacy evaluable population comprised 20 patients. The disease control rate was 100% (95% confidence interval [95% CI]: 83-100%) and the ORR was 95% (95% CI: 75-100%). At the time of follow-up cutoff, the best CRR reached 90% (95% CI: 68-99%). Out of these, only one patient demonstrated stable disease and was switched to the other regimen. A total of 126 courses of immunochemotherapy were administered, with a median number of six courses per patient. The follow-up time averaged 191 days (range, 31-345 days), and the estimated PFS rate was 100% at 12 months. After 6-8 courses of tislelizumab plus GemOx, ten subjects received tislelizumab maintenance, and all remain in remission so far, with the longest follow-up of 345 days. Overall, the treatment was well tolerated with the majority of adverse events (AEs) being grade 1-2 in severity. Serious AEs were grade 3 anemia (n=1) and grade 3 thrombocytopenia (n=1).
Conclusion
Tislelizumab plus GemOx demonstrated promising antitumor activity with manageable toxicities as a salvage treatment for RRcHL. A longer follow-up is needed to demonstrate the durability of the remission after tislelizumab maintenance.
No relevant conflicts of interest to declare.