Abstract
Background
Relapsed and refractory multiple myeloma (RRMM) remains a major challenge. With each relapse, patients (pts) experience decreased response duration leading to shortened survival. Pts with triple-class refractory disease (refractory to one class of the following: immunomodulatory agents (IMiDs), proteasome inhibitors (PI) and anti-CD38 monoclonal antibody) have a poor prognosis. Belantamab mafodotin is a first-in class B-cell maturation antigen (BCMA) antibody-drug conjugate. The aim of this study was to analyze the clinical outcomes of belantamab/dexamethasone (Bd) in triple-class RRMM.
Patients & Methods
Twenty-eight pts with triple-class RRMM receiving Bd were identified at University of Kansas Health System between October 2019 and June 2021 and reviewed retrospectively. These pts received belantamab 2.5 mg/kg IV every 3 weeks and dexamethasone (20-40) mg PO weekly. Descriptive analyses were performed on available data for patient characteristics. Survival curves were generated using the Kaplan-Maier method. Responses were evaluated using the International Myeloma Working Group (IMWG) criteria.
Results
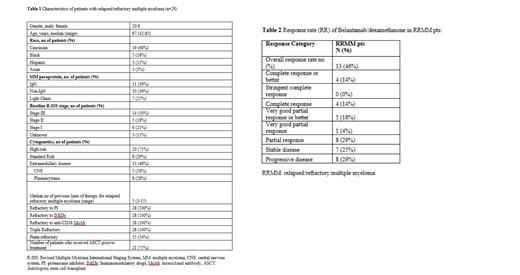
The median age was 67 yrs (42-85). Eleven pts (39%) had IgG isotype, 14 pts (50%) had R-ISS stage III disease, 20 pts (71%) had high-risk cytogenetics, and 13 pts (46%) had extramedullary disease (EMD). Patients characteristics are summarized in Table 1. Median number of Bd cycles received was 3 (2-18). The median number of previous lines of therapy was 5 (3-15). All pts were triple-class refractory, whereas 15 pts (54%) were penta-refractory. Twenty-one pts (75%) received autologous stem cell transplant, and 8 pts (29%) had previously received BCMA-targeted therapy. The response rate for all pts was 46% with 18% achieving very good partial response and better. Median follow-up was 7.4 months. Median progression-free survival (PFS) was 4.9 months, while median overall survival (OS) was 7.4 months. The response rates are summarized in Table 2.
Keratopathy was one of the most common adverse events (AEs), occurring in 23 (82%) pts, 13 (56%) pts had grade 3 or 4 keratopathy. Nineteen patients (68%) required dose reduction or delay due to keratopathy. Other common AEs included: anemia (83%), thrombocytopenia (70%), neutropenia (30%), and elevated liver function tests (53%). Eighteen patients (64%) discontinued due to progression of disease or death. No treatment-related mortality was noted in this review.
Conclusion
Our analysis demonstrates a reasonable efficacy of Bd in those who are heavily treated triple-class RRMM patients in the real world. Keratopathy remains a challenging AE and the main cause of dose reduction and delay.
Atrash: AMGEN: Research Funding; Jansen: Research Funding, Speakers Bureau; GSK: Research Funding. Mahmoudjafari: GSK: Membership on an entity's Board of Directors or advisory committees; Omeros: Membership on an entity's Board of Directors or advisory committees; Incyte: Membership on an entity's Board of Directors or advisory committees. McGuirk: Magenta Therapeutics: Consultancy, Honoraria, Research Funding; Allovir: Consultancy, Honoraria, Research Funding; Kite/ Gilead: Consultancy, Honoraria, Other: travel accommodations, expense, Kite a Gilead company, Research Funding, Speakers Bureau; Gamida Cell: Research Funding; Novartis: Research Funding; EcoR1 Capital: Consultancy; Juno Therapeutics: Consultancy, Honoraria, Research Funding; Novartis: Research Funding; Fresenius Biotech: Research Funding; Astelllas Pharma: Research Funding; Bellicum Pharmaceuticals: Research Funding; Pluristem Therapeutics: Research Funding.