Abstract
Immune thrombocytopenia (ITP) is an autoimmune disease characterized by decreased platelet (PLT) count in peripheral blood and marrow megakaryocyte dysfunction, which was induced by antiplatelet antibodies or cytotoxic T cells. Many ITP patient were previously full response to first-line treatment such as corticoids (CIS) or immunoglobulin (IVIG), but were intolerant or relapse during the maintenance treatment, and turn to receive recombinant human thrombopoietin (rhTPO), rituximab (RTX), splenectomy as well as immunosuppressive therapy (IST). Unfortunately, there still some patients failed to multiple treatments are faced with high bleeding risk.
Eltrombopag, the first oral non-peptide thrombopoietin receptor agonist (TPO-RA) that approved by US FDA in 2008, is the second-line treatment option for chronic ITP based on guideline recommendation. As reported, the effective rate of eltrombopag in ITP ranges from 59% to 89.7%, and limited predictive factors were available to assess the efficiency for individual. The characteristic of marrow megakaryocyte (MK) in ITP was reported as normal or hyper megakaryocytosis on the initiation (before treatment), however there are also ITP patients with decreased marrow MK, and the response of eltrombopag to these patients have not been fully reported. Bone marrow aspiration is a routine examination in China for the diagnosis of ITP with severe thrombocytopenia. Therefore, we conducted a research tried to assess the efficiency of eltrombopag to patients with multi-line failed ITP, and analyzing the possible factors may contribute to the differences based on personal characteristic.
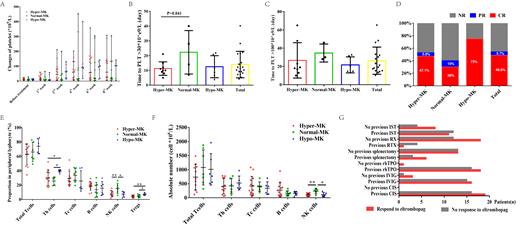
Thirty-five multi-line failed ITP patients with PLT count bellow 30× 10 9/L who received eltrombopag treatment were enrolled, and divided into three groups, the hyper-MK (MK count>35, n=17), normal-MK (8-35, n=10) and hypo-MK (≤7, n=8) groups. The average PLT count was 10.63±6.30×10 9/L before eltrombopag treatment, and it increased significantly to 12 (2-65)×10 9/L, 39 (2-212)×10 9/L, 68 (2-453)×10 9/L, 60 (3-358)×10 9/L, and 75 (3-308)×10 9/L in the 1st, 2nd, 4th, 6th , and 8th weeks after treatment (all P=0.000, except 1st week). The median intervals time required for platelet to reach 30×10 9/L and 100×10 9/L for the first time were 11 (4-40) and 21 (10-65) days, respectively (Fig. A-C). We found the overall, complete (PLT count ≥100×10 9/L) and partial response (PLT count ≥30×10 9/L or at least twice the baseline value) rates were 54.3% (n=19), 48.6% (n=17), and 5.7% (n=2) respectively to eltrombopag in our center. The overall response rate of patient with decreased MK was 75%, which was unexpectedly higher than the patient with increased or normal MK count (52.9% and 40%, respectively) (Fig. D). To explain the underling mechanism, we detected the peripheral T lymphocyte, B lymphocytes and NK cells before eltrombopag. Results showed that the patient with decreased MK were characteristic with higher T helper (Th) cells (39.62±2.01%) and regulatory T (Tregs) cells (7.93±1.63%) when compared to the hyper-MK (30.44±10.95%, 4.23±1.67%) and normal-MK group (25.67±5.72%, 4.81±2.12%). For patient with poorer eltrombopag response group, more percentage and absolute number of NK cells (15.48±7.12%, 234.4±91.80×10 6/L) were found in peripheral blood (Fig. E-F). We also discovered that previous first-line (CIS or IVIG) treatment, rhTPO effectiveness, RTX splenectomy and IST had no influence on eltrombopag response (Fig. G). As for safety, nine eltrombopag related adverse events were reported, and most commonly were upper respiratory tract infection (8.6%), elevated ALT (5.7%), and venous thrombosis (5.7%). All the side effect was cured by symptomatic treatment, eltrombopag dose reduction or discontinue.
In summary, ITP patients with decreased megakaryocyte respond well to eltrombopag, and the abnormality of NK cells may play a role in patients exert a poor response. In further clinical practice, we should considering the megakaryocytes count as well as peripheral NK population to better predicting eltrombopag response in ITP patients.
No relevant conflicts of interest to declare.