Abstract
Gene-modified hematopoietic stem cells (HSCs) therapy has demonstrated remarkable success for the treatment of inherited blood disorders. As the origin of hematologic hierarchy, HSCs play an essential role in sustaining life-long hematopoiesis. HSCs identification via reliable and robust bio-markers could facilitate the development of HSC gene therapy. Previous studies showed that long-term hematopoietic stem cells (LT-HSCs) were enriched in the Lin -CD34 +CD38 -CD45RA -CD90 +CD49f + population which could support long-term hematopoietic reconstitution. However, several of these surface markers proved to be unreliable when ex vivo culturing, such as CD38 and CD49f. Thus, HSCs characterization is still hindered by lacking bona-fide bio-markers, and consequently identification of long-term HSCs still needs time-consuming in vivo transplantation. To this end, we performed in vitro screening and comprehensive functional evaluation to identify a novel surface marker of human HSCs.
During initial screening, a cell surface antigen screen panel (including 242 human cell surface markers) and human CD34 and CD90 antibodies were used to perform flow cytometry analysis on CD34 + HSPCs enriched from umbilical cord blood. Compared with CD34 + cell population, we found that CD66 (a,c,d,e), CD200 and CD48 positive cells were more enriched in CD34 +CD90 + subset. Previous studies indicated that HSCs cannot be maintained during in vitro culturing. By tracking these candidate surface markers based on this principle, CD66e was selected as the potential HSCs bio-marker.
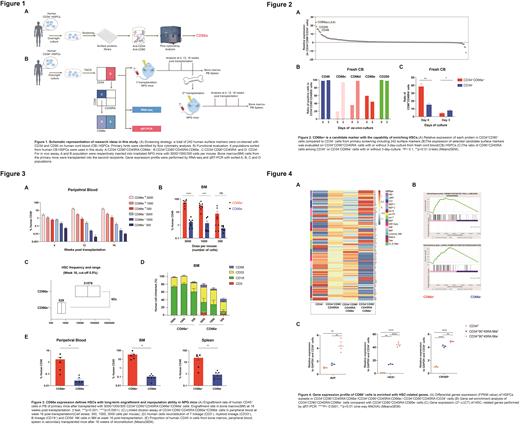
Next, we examined the in vivo hematologic repopulating potential of HSCs by limiting dilution assay (LDA) on immune-deficient mouse model. We sorted CD66e + and CD66e - subsets from CD34 +CD90 +CD45RA - subpopulation, and transplanted into irradiated NOD-scid Il2rg −/− (NPG) mice respectively. At week16 post-transplantation, in contrast to the CD66e - group, CD66e + cells exhibited significantly higher reconstitution in peripheral blood (PB), bone marrow (BM) and spleen. Engraftment dynamics revealed that the CD66e - group were only capable of reconstitution 4 weeks post transplantation, even at the highest initial cell dose. Moreover, the CD66e - group displayed impaired multi-lineage differentiation pattern, especially in PB and BM samples, while the CD66e + group presented a robust multi-lineage reconstitution. Notably, LDA results showed that the CD66e + cells within CD34 +CD90 +CD45RA - population contained 1 out of 529 SCID repopulating cells (SRC), almost 60-fold greater than the CD66e - fraction. To further investigate the long-term repopulating potential of the CD66e + cells, we performed the secondary transplantation collected from the BM cells of primary recipients. CD66e + cells presented significant higher repopulating activity than CD66e- subset in the secondary recipients. These findings reveal that the major cells with homing and long-term reconstitution capacity among CD34 +CD90 +CD45RA - cells were CD66e positive.
In order to determine the transcriptional profile of CD66e + cells, we performed RNA-sequencing analysis using the population of CD34 + cells, CD34 +CD90 +CD45RA - cells, CD66e + and CD66e - cells within CD34 +CD90 +CD45RA - subset. Remarkably, compared with other groups, the CD66e + cells displayed a bias toward the signature of HSC and early progenitors such as LMPP and CLP. Moreover, gene set enrichment analysis showed that hematopoietic lineage and long-term potentiation-related genes were highly enriched in the CD66e + cells. Further qRT-PCR experiment confirmed that several HSC-related genes were significantly higher expressed in CD34 +CD90 +CD45RA -CD66e + cells, compared to CD66e - population or CD34 + HSPCs, suggesting that the gene expression profile of CD66e + cells is reminiscent of HSC signature.
Altogether, we demonstrate that CD66e is a robust functional HSC bio-marker that CD66e-positive population among CD34 +CD90 +CD45RA - cells exhibit typical HSC signature, enhanced in vivo engraftment potential and robust multilineage differentiation pattern, which will provide an invaluable tool to investigate the origin of human HSCs, paving the way for the therapeutic application.
Fang: EdiGene, Inc.: Current Employment.