Abstract
Introduction Data on the effectiveness and safety of new vaccines against COVID-19 in patients (pts) with hematological diseases are just beginning to accumulate. We planned to obtain such information for pts with chronic myeloid leukemia (CML) during vaccination.
Objective. To evaluate the antibodies formation and adverse events (AEs) after vaccination against COVID-19 in pts with CML
Materials and methods. All pts with CML diagnosis who applied to the National Research Center for Hematology (NRCH, Moscow, Russia) for outpatient or remote consultations were suggested to prospectively report the AEs after getting a vaccination against COVID-19 by the most frequently used vector-based vaccine GamCovidVac (Sputnik V). Two vaccine components with the interval of 21 days were given at the vaccination facilities, as prescribed. At least after 3 weeks after the 2 nd injection, pts were advised to perform a blood test for the specific antibodies against spike (S) protein of SARS-CoV-2. A semi-quantitative test detecting the SARS-CoV-2 S1 subunit (RBD) IgG antibodies by enzyme-linked immunoassay (ELISA) kit was used in the clinic. The results were considered positive with the cutoff index >1,1. The use of any other lab tests detecting antibodies to S protein of SARS-CoV-2 was acceptable as well.
Results. In total, 66 pts with chronic phase of CML received a vaccination by Sputnik V in the 7 months period (from 18.12.2020 to 20.07.2021). Me age was 54 years (range 29 - 89 years), 34 (52%) were males. Median (Me) CML duration was 8 years (from the moment of diagnostics up to 20 years). Fifty one (77%) pt received TKI therapy and 15 (23%) were off-therapy at the time of vaccination, including 12 (18%) in a treatment-free remission and 3 (4,5%) pts in the process of diagnosis. Deep and major molecular response (MMR) was in 46 (70%) and 7 (11%) pts, respectively. Two (3%) pts had a molecular response MR2, 11 (17%) had no MR2. Eight (12%) pts had a history of COVID-19 manifestation prior to vaccination.
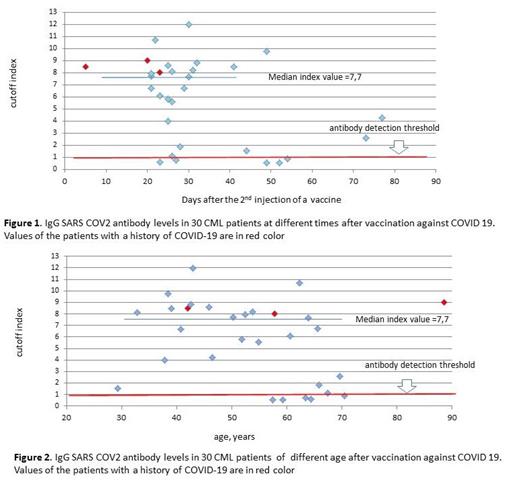
Me time for testing for the antibodies was 27 days (range 5-77) after the 2 nd vaccine injection. The tests were done in 44 (67%) of pts and revealed positive by any of the test systems in 42 (95%) pts. ELISA test was used in 30 (45%) pts and was positive in 25 (83%) of 30 pts. Me cutoff index in the positive samples was 7,7 (range 1,1 - 12) and corresponded to the value observed in healthy people after vaccination (medical stuff, data not shown). In all 3 pts with the history COVID 19, the index of positivity was above the Me value (Fig. 1, 2).
Other test systems were used in 14 (21%) pts, in all 14 (100%) the antibodies were found. In 3 of 5 patients with the cutoff index<1 the antibodies were detected by using other test systems, but all with a level slightly above the detection threshold. Me age of these 5 pts was 63 years (range 59- 70), Me time of analysis was 49 days (range 23-59) after 2 nd vaccine shot. All these pts were on treatment by tyrosine kinase inhibitors, 3 pts with MMR and deeper, 1 pt with MR2 and 1 pt without MR2.
A weak reverse correlation of the antibody levels with the time after vaccination was noted ( r = - 0,39, p = 0,033). A very weak reverse correlation with age was observed ( r = - 0,28, p = 0,127) (Fig. 1, 2).
No AEs after the vaccination were observed in 25 (38%) pts while 41 (62%) pts reported the AEs and 7 (10%) pts did not report their reactions. The AEs were as follows: local pain/discomfort in the injection site in 19 (29%) pts, weakness and/or drowsiness in 20 (30%), fever and/or chills in 16 (24%), other reactions in 8 (12%) including headache, heartbeat, lower back pain, pain in limbs, activation of herpes infection.
Conclusion: The single center study revealed no unusual or unexpected AEs in CML pts after the vaccination against COVID-19 by Sputnik V vaccine. The proportion of CML pts with specific antibodies after was 95% which is close to the published results of the 3rd phase study. No significant correlation was found with age (r = -0,28, p = 0,127), however, the absence or very low antibody levels were detected in individual patients aged about 60-70 years. This data raise a question of a necessity for a non-specific protection (masks, respirators, distance etc) and probably considering additional vaccination in some elderly persons. The duration of a humoral response against COVID-19, protective antibody titer and connection with clinical outcomes in CML pts need further evaluation in parallel with a common population.
Chelysheva: Pfizer: Speakers Bureau; Pharmstandart: Speakers Bureau; Bristol Myers Squibb: Speakers Bureau; Novartis Pharma: Speakers Bureau. Petrova: Pfizer: Speakers Bureau; Novartis Pharma: Speakers Bureau. Gurianova: Pfizer: Speakers Bureau. Turkina: Pharmstandart: Speakers Bureau; Pfizer: Speakers Bureau; Novartis Pharma: Speakers Bureau; Bristol Myers Squibb: Speakers Bureau.