Abstract
Vaso-occlusive events (VOEs) and pain crises are common clinical features of sickle cell disease (SCD) that result from sickle-shaped erythrocytes and leukocytes blocking blood flow, particularly in small vessels (Kato 2018). Activated endothelium also plays a role in the pathogenesis of VOEs in SCD. For example, VCAM-1 is expressed on blood vessels after activation by chemical and/or mechanical stimulation, which results in cytokine release. Studies have shown that patients with SCD have higher steady-state serum levels of soluble VCAM-1 compared to controls and that these levels are elevated during VOEs (White 2020). Furthermore, overexpression of these adhesion molecules on the endothelium results in prolonged adherence of white blood cells (WBCs), which has been shown to contribute to the development of VOEs and possibly cerebral vasculopathy. These findings raise the need to explore further the role of aberrant WBC- and/or RBC-endothelial interaction, mediated via VCAM-1, in the pathophysiology of cerebral microvascular hemodynamics and vasculopathy leading to cerebral microinfarcts in SCD. Therefore, we hypothesized that sickle cell mice will show greater cerebral cortical expression of VCAM-1 compared with age-matched controls and that this deposition will be associated with significant evidence of abnormal cerebral microvascular hemodynamic abnormalities.
To examine the relationship between abnormalities in cerebral microvascular hemodynamics and VCAM-1 deposition in the cerebral microvasculature, we utilized a humanized sickle cell (with HbSS) and corresponding control (with HbAA). After cranial-window procedures, cortical capillaries, precapillary arterioles, and post-capillary venules were imaged using two-photon microscopy at two time points. In addition, this experiment included pre-and post-transfusion groups as we intend to study the impact of blood transfusion on hemodynamics. Using custom-written but well-validated MATLAB scripts, we analyzed line scans to identify the number and duration of rolling or adherent WBCs and RBCs, the RBC velocity in cerebral microvasculature, and the frequency and magnitude (mL/sec) of cerebral microvascular blood flow reversals. Rolling WBCs were defined as lasting two seconds or more, and adherent RBCs were defined as lasting 0.5 seconds or more. To quantify the expression of VCAM-1, we used immunohistochemistry to stain 50-micron sections of brain tissue for VCAM-1, Lectin to localize the vasculature, and Neun to localize neuronal nuclei. Images were analyzed using Phenochart and ImageJ software to examine the deposition of VCAM-1 throughout the brain tissue.
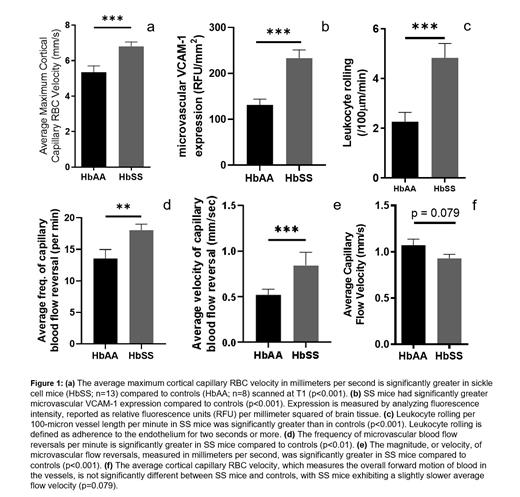
As shown in Figure 1, at the first time point (baseline), we observed a significantly higher maximum RBC velocity (p<0.001) in the sickle cell mice compared to controls (figure 1a). We also found that there was significantly higher expression of VCAM-1 (p<0.001) (figure 1b) as well as significantly more leukocyte rolling (p<0.001) (figure 1c) in the sickle cell mice compared to controls. Additionally, we noted that the sickle cell mice have a significantly higher frequency of blood flow reversals (p<0.01) (figure 1d) as well as higher magnitude of microvascular blood flow reversals (p<0.001) (figure 1e) compared to controls. Interestingly, the sickle cell mice have a slightly lower average or mean capillary blood flow velocity compared to control (figure 1f), but this was not statistically significant (p=0.079). Since the mean capillary velocity is obtained as a smoothened difference between the forward flow and reversals, this decrease was surprising given the significant differences in frequency and magnitude of microvascular blood flow reversal in the sickle cell mice compared to controls (figs 1d and 1e).
In conclusion, we see that the high velocity of blood flow might be a mechanical force, among other factors contributing to cerebral microvascular VCAM-1 expression in sickle cell mice. This might be responsible for the increased leukocyte-endothelial interactions and adhesion, ultimately leading to higher frequency and magnitude of cerebral microvascular blood flow reversal. Taken together, this may contribute to the observed slightly lower mean or effective microvascular forward blood flow. These pathophysiological changes might contribute to the reported higher rate of cerebral microinfarct and silent infarct in sickle cell disease.
Archer: Global Blood Therapeutics: Consultancy, Research Funding; Forma Therapeutics: Research Funding. Hyacinth: Novartis: Consultancy; Acuta Capital: Consultancy.