Niche cells within the bone marrow (BM) microenvironment support the maintenance and reconstitution of hematopoiesis. In this issue of Blood, Yu et al demonstrate that angiopoietin-like 2 (ANGPTL2) secreted by the vascular niche endothelial cells serves as the seminal paracrine angiocrine factor, orchestrating hematopoietic stem cell (HSC) homeostasis and regeneration after myelosuppression.1
Tissue-specific endothelium, such as BM endothelial cells (BMECs), is endowed with unique organotypic specialized functions, including modulation of metabolism, immune cell trafficking, inflammatory response, vascular tone, regeneration, and repair.2,3 It is well documented that blood vessels in the BM are not just passive conduits delivering nutrients and oxygen to hematopoietic cells. BMECs execute specialized hematopoietic-supportive functions that promote hematopoietic stem and progenitor cell (HSPC) proliferation and self-renewal through a coordinated angiocrine supply of stimulatory and inhibitory membrane-bound and secreted factors.4,5 Direct cellular contact between BMECs and hematopoietic cells is essential for balanced self-renewal and differentiation of HSCs and HSPCs6 in homeostatic and regenerative states (see figure). Although numerous angiocrine factors, including bone morphogenetic proteins (BMP2 and BMP4), Notch ligands, CXCL12, and Kit ligand, could be supplied by other BM niche cells, certain factors presented specifically by BMECs might exert dominant functions in HSC homeostasis. However, the identity of these unique vascular niche–derived HSC-dominant angiocrine factors are not well defined. Notably, growth factors, such as ANGPTL2, which have been shown to regulate HSC metabolic, inflammatory, and angiogenic functions,7 could also be secreted by diverse BM niches cells, including BMECs, stromal cells, and hematopoietic cells themselves. Whether preferential secretion of ANGPTL2 by a specific niche cell drives hematopoiesis has not been formally studied.
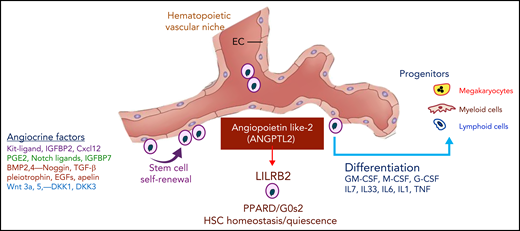
The balanced supply of stimulatory and inhibitory angiocrine factors choreograph the self-renewal and differentiation of HSCs and HSPCs. Secretion of ANGPTL2 specifically by vascular niche endothelial cells within the hematopoietic microenvironment supports the maintenance of HSCs and recovery from myeloablation. ANGPTL2, through interaction with leukocyte immunoglobulin-like receptor B2 (LILRB2) expressed on HSCs, switches on the expression of PPARD and G0s2, thereby dictating the homeostatic equilibrium of HSCs. G-CSF, granulocyte colony-stimulating factor; GM-CSF, granulocyte-macrophage colony-stimulating factor; IL-7, interleukin 7; M-CSF, macrophage colony-stimulating factor; TGF β, transforming growth factor β; TNF, tumor necrosis factor.
The balanced supply of stimulatory and inhibitory angiocrine factors choreograph the self-renewal and differentiation of HSCs and HSPCs. Secretion of ANGPTL2 specifically by vascular niche endothelial cells within the hematopoietic microenvironment supports the maintenance of HSCs and recovery from myeloablation. ANGPTL2, through interaction with leukocyte immunoglobulin-like receptor B2 (LILRB2) expressed on HSCs, switches on the expression of PPARD and G0s2, thereby dictating the homeostatic equilibrium of HSCs. G-CSF, granulocyte colony-stimulating factor; GM-CSF, granulocyte-macrophage colony-stimulating factor; IL-7, interleukin 7; M-CSF, macrophage colony-stimulating factor; TGF β, transforming growth factor β; TNF, tumor necrosis factor.
To address this, Yu et al used sophisticated gene knockdown in each of the niches to demonstrate that angiocrine-derived ANGPTL2 selectively secreted by BMECs, but not other stromal niche cells, serves as a critical factor that maintains and re-establishes hematopoietic cell stemness. To demonstrate the specific angiocrine ANGPTL2 function, Yu et al leveraged a multiprong lineage-specific strategy in mice, whereby the Angptl2 gene was silenced in different niche components, including vascular cells, mesenchymal cells, and megakaryocytes, to provide a unique insight into how niche-specific factors determine the biological behavior of HSCs. Although other studies have demonstrated a functional overlap among mesenchymal, osteoblastic, perivascular, and vascular niches, Yu et al revealed the cardinal role of only endothelial cell–derived ANGPTL2 in maintaining HSC functions at steady-state conditions and during hematopoietic recovery following 5-fluorouracil–induced hematopoietic ablation. ANGPTL2 deletion in BMECs, but not in stromal cells, resulted in impairment of HSC sustenance and reconstitution after hematopoietic ablation.
Mechanistically, Yu et al showed that, through activation of its receptor human leukocyte immunoglobulin-like receptor B2,8 ANGPTL2 enhances peroxisome proliferator activated receptor δ (PPARD) expression to directly transactivate G0/G1 switch 2 (G0s2) levels to maintain the quiescent status of HSCs (see figure). PPARs are nuclear receptors that play pivotal roles in the regulation of the metabolic status of various cell lineages. The most dramatic change observed in the BM niche during aging is excessive adipocyte expansion that presumably has a negative impact on HSC function.9 An altered metabolic microenvironment in the stressed and aged BM might induce the defects observed in HSCs, setting the stage for myelodysplasia and hematopoietic failure during recovery from myeloablation. Because endothelial cells act as critical gatekeepers of tissue-specific metabolites, including free fatty acids, it is plausible that imbalanced metabolic fluxes and inflammatory stress within BMECs can result in aging-induced damage within the BM niche, thereby impairing HSC functions. Based on the result illustrated in the article by Yu et al, aberrant secretion of endothelial cell–derived ANGPTL2 might instigate metabolic disruptions that contribute to HSC dysfunctions manifested in myelodysplastic maladies, induction of clonal hematopoiesis, and aging, as well.
Within the hematopoietic microenvironment, BMECs manifest remarkable intraorgan vascular heterogeneity,2,3 in which specialized arterial, sinusoidal, and periosteal endothelial cells provide unique temporal and spatial complexity, modulating HSC quiescence and proliferation during hematopoietic development, steady-state conditions, and regeneration from myelosuppression. Yu et al show that, within the BM, ANGPTL2 could potentially be expressed by almost all of the specialized endothelial cell subtypes. Whether differential expression of ANGPTL2 specifically by arterial, periosteal, or sinusoidal vessels selectively enforces the quiescence or proliferative self-renewal of HSCs was not studied and needs to be investigated further. In addition, it is not obvious whether ANGPTL2 has a preference for differential expansion of particular lineage-specific hematopoietic cells or primarily drives hematopoiesis by solely modulating HSC stochastic differentiation into myeloid and lymphoid lineages.
Although the selective knockdown of ANGPTL2 by various niche cells clearly shows that its secretion exclusively by BMECs is essential for its biological activity, it remains to be determined whether ANGPTL2 might also affect the prohematopoietic functions of other niche cells, including adipocytes, mesenchymal cells, and osteoblasts. Specifically, through interactions with its other putative receptor, CD146,10 that is expressed on subsets of the stromal cells, ANGPTL2 could mastermind hematopoietic recovery. Notwithstanding these caveats, Yu et al raise the intriguing possibility that compartmentalization of angiocrine factors, primarily by the vascular niche, could also assign various niche cells to different roles in controlling hematopoietic functions.
The findings by Yu et al could open the door for selective targeting of the individual niche cells for therapeutic hematopoietic reconstitution. For example, in certain hematopoietic failure states, selective infusion of healthy BMECs capable of homing to the BM microenvironment and secretion of physiological levels of ANGPTL2 could mitigate injury to hematopoietic cells caused by chemotherapy, radiation, or immunotherapy. Furthermore, the evidence furnished by Yu et al might justify exploration of clinical approaches in which infusion of ANGPTL2 protein by itself might restore hematopoietic recovery after myeloablation.
Conflict-of-interest disclosure: The authors declare no competing financial interests.