Abstract
INTRODUCTION Diffuse Large B-cell Lymphoma (DLBCL) is most frequently diagnosed in the elderly, with a median age of presentation of 66 years. While approximately 60% of patients can achieve a cure with intensive anthracycline - based chemoimmunotherapy, treatment is associated with toxicity and may not always be safe in frail or elderly patients. Little is known about characteristics and outcomes in patients who are too frail for treatment or opt to not receive systemic therapy.
METHODS: We used the Surveillance, Epidemiology, and End Results Program (SEER) linked with Medicare claims to identify 6,214 DLBCL patients (pts) diagnosed between 2007 and 2017. Data collected included demographic variables; comorbidity burden calculated using the Elixhauser comorbidity index with conditions present 30 to 365 days prior to DLBCL diagnosis. Initial therapy was defined as prescribed -30 to 100 days post-diagnosis. Patients who did not receive outpatient treatment but had current procedural terminology (CPT) codes reflecting inpatient chemotherapy were excluded from analysis.
RESULTS: Of the 6,214 patients, 1,644 (26.5%) were untreated. Untreated patients were older with a median age of 81 years (interquartile range (IQR) 74-87) compared to 78 years old (IQR: 72-84) in the total cohort. Patients 85 years and older (≥85 y) represented 22.0% of the total cohort, 36.2% of untreated patients and 13.8% of patients receiving treatment for DLBCL. Overall, 43.6% of patients ≥85 y did not receive treatment. This percentage also varied by race: Of 194 black patients, 33% were not treated, compared to 26.4% of white patients.
During the study period, 60.7% of the total cohort died. The proportion of deaths was higher in the untreated cohort (83.7%) and lower (45.4%) in the chemotherapy-treated cohort. Median survival of untreated patients was 3 months (IQR 2-27) . The median survival of the total cohort was 18 months (IQR 4-47).
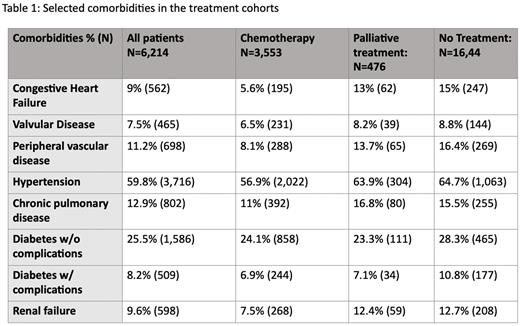
The comorbidity burden was higher in the untreated patients compared to both the total cohort and chemotherapy treated patients: 15% had congestive heart failure (CHF), 16.4% had peripheral vascular disease, and 10.8% had diabetes with complications. Table 1 lists selected Elixhauser comorbidities at time of diagnosis. Of 562 DLBCL patents with a diagnosis of CHF, 44.0% did not receive any treatment.
CONCLUSIONS: Approximately one quarter of DLBCL patients in the SEER - Medicare database are listed as not receiving treatment. Untreated patients were older and had a higher burden of baseline comorbidities. Black patients were more likely to receive no treatment than white patients. Expectedly, untreated patients had short overall survival, highlighting the continued need for new tolerable therapies for DLBCL. Future analysis will focus on lymphoma specific mortality, time to event analyses and identification of populations at highest risk of not receiving therapy. These populations should be the target of research of novel therapeutic strategies in order to improve outcomes with minimized toxicity.
Disclosures
Caimi:Novartis: Membership on an entity's Board of Directors or advisory committees; Kite Pharma: Membership on an entity's Board of Directors or advisory committees; MEI Pharma: Membership on an entity's Board of Directors or advisory committees; Bristol Myers Squibb: Membership on an entity's Board of Directors or advisory committees; Takeda: Membership on an entity's Board of Directors or advisory committees; Genentech: Membership on an entity's Board of Directors or advisory committees; ADC Therapeutics: Membership on an entity's Board of Directors or advisory committees; Genmab: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.