Abstract
Follicular lymphoma (FL) is a clinically heterogeneous B cell non-Hodgkin lymphoma (B-NHL) subtype. While some patients (pts) tolerate watch-and-wait for years, others require prompt treatment. Further, a subset of pts suffers progression of disease within 24 months (mo) of treatment initiation (POD24) and/or aggressive histologic transformation (tFL). No predictive model guides FL treatment, but the FLIPI stratifies pt outcomes using clinical parameters. The m7-FLIPI integrates FLIPI, performance status (PS), and mutations in EZH2, ARID1A, MEF2B, EP300, FOXO1, CREBBP, and CARD11; however, it is not routinely used to guide treatment due to unproven generalizability and actionability to date.
We conducted a clinicogenomic analysis of 343 pts with FL or tFL [enrolled on CALGB/Alliance trials 50402, 50701, or 50803 (n = 58); or real-world cohorts from WUSTL (n = 191), Cleveland Clinic (n = 77), and U Miami (n = 17)], applying a custom 2,611-gene sequencing panel to B-NHL specimens. We explored patterns of mutated genes in newly diagnosed FL (n = 231), relapsed (rel) FL (n = 65), and tFL (n = 47). We also asked whether specific genomic features were associated with clinical parameters and assessed the association of different mutations with pt outcomes. Median age was 60 years (range: 22-89); stage distribution was 23% early vs 77% advanced; FLIPI distribution was 34% low vs 34% intermediate vs 32% high. Progression-free and overall survival (PFS/OS) of pts in our cohort exhibited anticipated relationships with key parameters (e.g., histology: 48-mo OS 96% vs 69% for FL vs tFL).
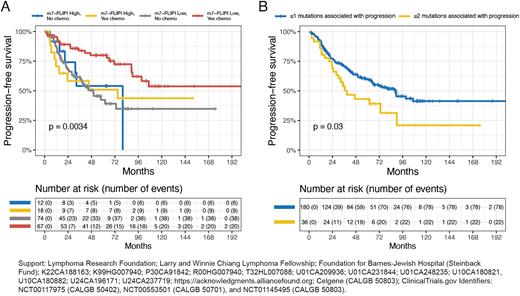
We applied the m7-FLIPI to our 231 newly diagnosed FL samples, observing weak prognostic impact [low vs high risk; HR for PFS 1.37 (95% CI 0.43-1.23), p = 0.2]. Given heterogeneous treatment histories of our pts compared to uniform pt treatment in the initial m7-FLIPI cohort, we tested for an interaction between m7-FLIPI and frontline treatment. We observed a strong interaction between m7-FLIPI risk stratification and receipt of cytotoxic chemotherapy (p < 0.01; Fig 1A). 48-mo PFS was 80% for pts with low risk m7-FLIPI receiving chemo-based frontline treatment, vs 50-55% for all three other pt subsets (high risk m7-FLIPI pts irrespective of chemo-status, and low risk m7-FLIPI pts receiving chemo-free treatment). This suggests that the prognostic value of the m7-FLIPI mutational pattern is impacted by pt treatment.
To probe the links between the B-NHL genomic landscape and pathologic or clinical features, we compared the mutation burden (median number of mutations) across a range of B-NHL subsets. FL cohorts based on grade 1-2 vs 3A, early vs advanced stage, FLIPI, initial treatment vs observation, and POD24 vs no POD24 did not differ by mutation burden. However, mutation burden was significantly higher (p < 0.01) in tFL than rel FL, and in rel FL than newly diagnosed FL. Using a Cox proportional hazard penalized splines model, we did not find a significant relationship between mutation burden at FL diagnosis and PFS. CREBBP was the only gene in which mutations were significantly more common in FL than tFL (BH FDR q < 0.1), yet mutated CREBBP was associated with shorter frontline PFS in pts with FL (p < 0.05). Conversely, mutations in 23 other genes were more common in either rel FL or tFL compared to newly diagnosed FL (BH FDR q < 0.1), including 6 significantly mutated genes (SMGs; by MutSigCV): STAT6, TP53, IGLL5, B2M, SOCS1, and MYD88. We hypothesized that pts with newly diagnosed FL may be at high risk of poor outcomes if their pre-treatment tumors harbor mutations more common in progressive/transformed disease. We defined a 'mutations associated with progression' (MAP) signature as 2 or more mutations in these 7 genes (6 SMGs enriched in progressive/transformed disease and CREBBP). Applying univariate and multivariate Cox proportional hazard modeling (adjusting for FLIPI and PS), pts with newly diagnosed FL possessing a MAP signature had significantly shorter frontline PFS [multivariate HR for PFS 1.9 (95% CI 1.12-3.2), p < 0.05; median PFS 39 vs 89 mo, p < 0.05; Fig 1B]. In summary, our analyses highlight a set of MAPs that may offer unique insight into progression risk in FL, which may be more generalizable than the m7-FLIPI. Future studies are warranted to validate the poor prognosis associated with the presence of MAPs in pts with untreated FL, which could facilitate prospective studies of novel therapies in this subset of high risk pts.
Disclosures
Mehta-Shah:Daiichi Sankyo: Membership on an entity's Board of Directors or advisory committees, Research Funding; Genetech/Roch: Membership on an entity's Board of Directors or advisory committees, Research Funding; Bristol Myers-Squibb: Research Funding; AstraZeneca: Consultancy, Membership on an entity's Board of Directors or advisory committees; Kyowa Hakko Kirin Co., Ltd.: Membership on an entity's Board of Directors or advisory committees; Karyopharm Therapeutics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Celgene: Research Funding; Corvus Pharmaceuticals: Research Funding; Secura Bio: Membership on an entity's Board of Directors or advisory committees, Research Funding; Verastem: Research Funding. Kahl:AstraZeneca: Consultancy, Research Funding; Roche: Consultancy; Pharmacyclics: Consultancy; AcertaPharma: Consultancy; MEI: Consultancy; Abbvie: Consultancy, Research Funding; Genentech: Consultancy, Research Funding; Beigene: Consultancy, Research Funding; Kite: Consultancy; ADT Therapeutics: Consultancy; Celgene/BMS: Consultancy, Research Funding; Janssen: Consultancy; Incyte: Consultancy; Hutchmed: Consultancy, Research Funding; TG Therapeutics: Consultancy; Genmab: Consultancy; Seattle Genetics: Consultancy; Research To Practice: Speakers Bureau. Bartlett:Autolus, Bristol-Meyers Squibb, Celgene, Forty Seven, Janssen, Kite Pharma, Merck, Millennium, Pharmacyclics: Research Funding; Washington University School of Medicine: Current Employment; ADC Therapeutics, Roche/Genentech, Seattle Genetics: Membership on an entity's Board of Directors or advisory committees, Research Funding. Lossos:LRF: Membership on an entity's Board of Directors or advisory committees; Adaptive: Honoraria; NCI: Research Funding. Hsi:Virtuoso: Research Funding; Astellas: Consultancy; Cytomx: Consultancy; Novartis: Consultancy; Abcon: Consultancy; Abcon: Current holder of stock options in a privately-held company; Abbvie: Research Funding; Eli Lilly: Research Funding. Martin:ADCT: Consultancy; AstraZeneca: Consultancy; Beigene: Consultancy; BMS: Consultancy; Daiichi Sankyo: Consultancy; Epizyme: Consultancy; Genentech: Consultancy; Janssen: Consultancy; Regeneron: Consultancy; Takeda: Consultancy. Leonard:Roche/Genentech: Consultancy; Grail: Consultancy; Mustang Bio: Consultancy; Novartis: Consultancy; Merck: Consultancy; Eisai: Consultancy; Epizyme: Consultancy; Gilead/Kite: Consultancy; Genmab: Consultancy; Constellation: Consultancy; Calithera: Consultancy; Janssen: Consultancy; Incyte: Consultancy; Karyopharm: Consultancy; Lilly: Consultancy; MEI Pharma: Consultancy; BMS/Celgene: Consultancy; Pfizer: Consultancy; Seattle Genetics: Consultancy; Second Genome: Consultancy; Sutro: Consultancy; ADC Therapeutics: Consultancy; Bristol-Myers Squibb: Consultancy; Regeneron: Consultancy; Beigene: Consultancy; Miltenyi: Consultancy; Bayer: Consultancy; AstraZeneca: Consultancy; Astellas: Consultancy; AbbVie: Consultancy. Fehniger:HCW Biologics: Research Funding; ImmunityBio: Research Funding; BMS: Other: Provision of drug for investigator initiated trial; Indapta: Current holder of stock options in a privately-held company, Other: Scientific Advisory Board; Orca Bio: Current holder of stock options in a privately-held company; Wugen: Consultancy, Current holder of stock options in a privately-held company, Patents & Royalties; Affimed: Other: Scientific Advisory Board, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.