Abstract
Background At Boston Medical Center, patients across disciplines requiring IV iron are referred to a hematologist in the oncology infusion center. The high demand for patient appointments resulted in wait times up to 8 weeks from referral to visit across all hematologic indications. In addition, the absence of a standardized procedure led to increased drug denials, lack of reimbursement, treatment delays, and increased chair time. Pharmacist run clinics are becoming increasingly common and have offset time for general practitioners, improve access, and increase patient satisfaction. This quality improvement (QI) project aimed to increase the number of patients with iron deficiency anemia (IDA) managed by a pharmacist to improve physician access.
Methods Patients 18 years of age and older requiring IV iron for IDA were referred for management under a pharmacy run protocol in an outpatient clinic. Providers placed referrals through the electronic medical record (EMR), Epic, that were reviewed daily by pharmacists. Key inclusion criteria were established IDA inadequately controlled by oral iron due to poor absorption, inability to take oral iron, or requirement of rapid replenishment of iron stores. Under a scope of practice with hematologists, pharmacists assessed symptoms of IDA, ordered and interpreted iron laboratory results, selected appropriate IV iron formulation, created a treatment plan within the EMR, and scheduled follow-up. A standard operating procedure (SOP) was developed outlining a treatment algorithm with all available IV iron. Standard documentation was utilized and documented in the hospital EMR for each visit. Per the IHI model, Plan-Do-Study-Act (PDSA) cycles were implemented to assess change over time. PDSA cycle 1 was implementation of a pilot clinic for referrals restricted to hematologists. Outcome measures included total number of referred patients and provider time offset. Process metrics included percentage of visits with appropriate labs as outlined in the SOP. Balancing metrics included number of infusion-related reactions, incidence of hypophosphatemia, and number of pharmacist hours.
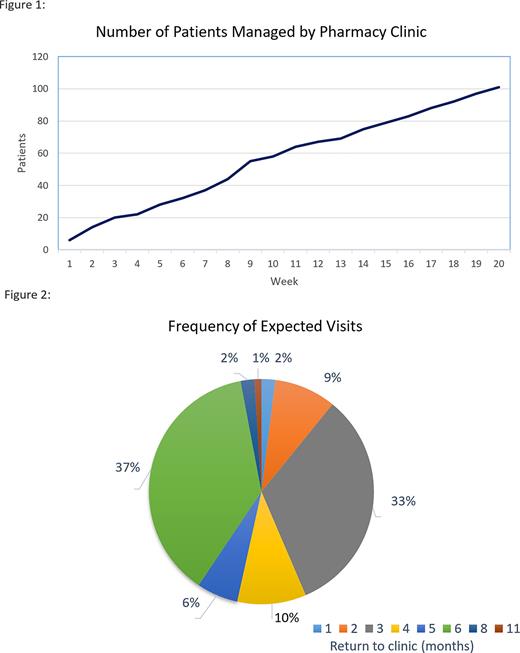
Results A total of 109 patients were referred to the pharmacy service from February 1, 2022 through June 17, 2022. Of 109 patients referred, 101 (92.6%) patients met inclusion criteria with an average of 5 patients referred weekly (Figure 1). Eight patients (7.3%) were excluded due to: complicated IDA that required further physician workup (4/8, 50%), the need to establish hematology care per the pilot protocol (2/8, 25%), adequate control on oral iron without previous IV therapy (1/8, 12.5%), or the absence of IDA (1, 12.5%). By June 17th, a total of 36 patients (35.6%) had an encounter with a pharmacist. The remaining patients (N=65) received an iron infusion at time of referral and did not require immediate follow-up. Time to next available appointment for patients requiring IV iron was < 7 days. Of patients seen by a pharmacist (N=36), 100% of patients had all per protocol iron studies completed. Six (16%) patients had phosphorus collected during their visit. Hypophosphatemia (phosphate < 2.7 mg/dL) was reported in 3 patients at time of initial pharmacist visit. There were no infusion-related reactions reported or documented in the patients evaluated. The total time spent by a pharmacist on all aspects of the protocol was 51.7 hours since clinic go-live for an average of 2.6 hours per week. More than 150 visits were projected to offset provider visits within 6 months based on the return to clinic time at referral, with a large majority of patients requiring follow-up at 6 months (37%, 37/101) or 3 months (33%, 33/101) (figure 2). Using the average reimbursement for an ambulatory oncology pharmacist visit, it was projected that this service would generate approximately $22,200 in visit revenue during the 6-month time period.
Conclusion The pharmacist driven iron clinic was successfully implemented in our ambulatory infusion center. Pharmacists served as an ancillary service under a scope of practice to help improve clinic efficiency by standardizing IV iron and facilitating timely access for patients requiring treatment. Planned PDSA cycles include expansion of referring providers to general internal medicine practitioners. This QI project will continue to streamline workflow for patients with IDA and will aid in future development of similar pharmacist driven practices.
Disclosures
Freydman:Pharmacosmos: Research Funding. AlDoughaim:Pharmacosmos: Research Funding. Gorelikov:Pharmacosmos: Research Funding. Tsou:Pharmacosmos: Research Funding. Sloan:Nuvectis: Consultancy; Abbvie: Consultancy; Stemline: Consultancy. Jhaveri:Pharmacosmos: Research Funding. Hughes:Pfizer: Current Employment; Abbvie: Speakers Bureau; Astrazeneca: Speakers Bureau; Bristol Myers Squibb: Consultancy; Karyopharm: Speakers Bureau; Rigel Pharmaceuticals: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.