Abstract
Background: Given the activity of allogeneic T cells in acute myeloid leukemia (AML) patients, immunotherapies stimulating antitumor T cell responses have been explored as a promising therapeutic option. However, these therapies have been associated with modest clinical activity, and further research is needed to identify response and resistance mechanisms. A spectrum of primitive and differentiated cells comprise AML in each individual patient, and this cell composition has been previously shown to predict outcomes. In particular, high proportions of AML stem and progenitor cells have been associated with worse clinical outcomes and lack of response to induction chemotherapy (Ng et al Nature 2016; van Galen et al Cell 2019; Zeng et al Nat Med 2022). The composition of AML cell differentiation states in relation to immunotherapy response or resistance has not been elucidated; herein, we explored whether this composition may be associated with response in AML patients treated with immune checkpoint blockade.
Methods: Pre- and posttreatment whole bone marrow (BM) samples were collected from 31 newly diagnosed and relapsed/refractory AML patients treated with azacitidine and pembrolizumab (NCT02845297) and 18 relapsed/refractory AML patients treated with high-dose cytarabine (HiDAC) and pembrolizumab (NCT02768792; Zeidner et al Blood Cancer Discov 2022) on two separate phase II trials. Primary patient samples and associated clinical data were obtained via informed consent in accordance with the Declaration of Helsinki as per research protocols approved by the Institutional Review Boards of the participating institutions. Bulk RNA sequencing was performed on all BM samples. Differential gene expression analysis was performed using the DESeq2 package, and gene set enrichment analysis (GSEA) was performed using the clusterProfiler package. Proportions of AML cell populations present in BM samples were deconvoluted using CIBERSORTx as per Zeng et al Nat Med 2022. The LSC17 score and LinClass7 score were calculated as per Ng et al Nature 2016 and Zeng et al Nat Med 2022, respectively. All analyses and graphs were generated using R (v4.2.1).
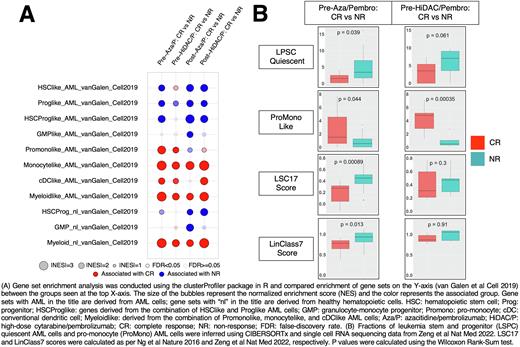
Results: GSEA using gene sets from a range of primitive and differentiated AML and normal cell types as per van Galen et al Cell 2019 was performed on pre- and posttreatment samples. Patients achieving a complete response (CR) to either immunotherapy regimen were enriched at baseline for the following differentiated AML gene sets: pro monocyte (Promono)-like AML; monocyte-like AML; conventional dendritic cell (cDC)-like AML; and myeloid-like AML (Figure 1A). Patients not responding (NR) to azacitidine/pembrolizumab were enriched for the following stem and progenitor AML gene sets: hematopoietic stem cell (HSC)-like AML; progenitor (Prog)-like AML; and HSC progenitor (HSCProg)-like AML (Figure 1A). To validate these findings, BM samples were deconvoluted into inferred fractions of AML differentiation states using CIBERSORTx and single-cell RNA sequencing expression data from Zeng et al Nat Med 2022. Patients not responding to azacitidine/pembrolizumab had a higher proportion of leukemia stem and progenitor quiescent cells (LSPC Quiescent) at baseline, while patients achieving CR to either regimen had higher baseline proportions of pro monocyte-like AML cells (Figure 1B). Additionally, patients not responding to azacitidine/pembrolizumab had higher baseline LSC17 and LinClass7 scores, two gene expression scores that are reflective of leukemia stemness (Figure 1B). In the posttreatment setting for both regimens, HSC-like, Prog-like, and HSCProg-like gene sets were associated with NR; monocyte-like and myeloid-like gene sets were associated with CR (Figure 1A).
Discussion: Patients achieving CR to azacitidine/pembrolizumab and HiDAC/pembrolizumab had higher proportions of differentiated AML cells, while NR patients had a higher proportion of AML stem and progenitor cells. We hypothesize that antitumor T cells may preferentially target differentiated AML cells over AML stem and progenitor cells. Further research is needed to validate these findings in a prospective and mechanistic manner and in other immunotherapy regimens. Analyses of the proportions of AML differentiation states in patients relapsing after allogeneic hematopoietic cell transplantation are ongoing.
Disclosures
Rutella:Wugen: Research Funding; MacroGenics: Research Funding; NanoString Technologies: Research Funding. Zeidner:AbbVie: Consultancy, Honoraria, Research Funding; Bristol Myers Squibb: Honoraria; Immunogen: Honoraria; Servier: Consultancy, Honoraria; Shattuck Labs: Honoraria; Arog: Research Funding; Astex: Research Funding; Jazz: Research Funding; Merck: Research Funding; Stemline: Research Funding; Sumitomo Dainippon Pharma: Research Funding; Syndax: Research Funding; Takeda: Research Funding; Genentech: Honoraria; Gilead: Consultancy, Honoraria, Research Funding. Serody:STING activation: Patents & Royalties: provisional patent to enhance CAR therapy for solid tumors/ provisional patent for the use of ILC2 cells to treat or prevent GvHD. Gojo:Ono Pharmaceutical: Membership on an entity's Board of Directors or advisory committees; Clearview: Membership on an entity's Board of Directors or advisory committees; Certara: Membership on an entity's Board of Directors or advisory committees; BMS: Membership on an entity's Board of Directors or advisory committees; Immunogen: Membership on an entity's Board of Directors or advisory committees; Celgene: Other: Research support; Genentech: Other: Research support; Gilead: Membership on an entity's Board of Directors or advisory committees, Other: Research support; Amphivena: Other: Research Support; Merck: Other: Research support; Amgen: Membership on an entity's Board of Directors or advisory committees, Other: Research Support; Johnson & Johnson: Membership on an entity's Board of Directors or advisory committees. Luznik:WindMil Therapeutics: Patents & Royalties; Genentech: Other: Clinical Trial Support, Research Funding; Gilead: Consultancy, Membership on an entity's Board of Directors or advisory committees; Rubius Therapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees; Talaris Therapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.