Abstract
Background: Patients with Cutaneous T Cell Lymphoma (CTCL) subtypes Mycosis Fungoides and Sézary Syndrome (MF/SS) who progress to advanced stage disease have a 20% 5-year overall survival rate. Current treatments for CTCL range from localized therapies for skin lesions to systemic treatments, including histone deacetylase inhibitors, chemotherapy, and immunotherapy. While many treatments are initially effective, patients often develop resistance to ongoing treatment. Though recent studies have comprehensively profiled the genomic landscape of CTCL, no clear driver mutation has been identified and the molecular factors that drive MF/SS progression remain undefined. To address these outstanding questions, we collected longitudinal tumor and normal samples from 10 MF/SS patients and subjected them to high resolution genome and transcriptome analyses (exome, whole genome and single-cell RNA sequencing, eWGS and scRNAseq) to define the clonal evolution and temporal progression of CTCL.
Methods: We collected 52 serial tumor samples (peripheral blood, skin and lymph node biopsies, 49-1050 days apart) and matched, purified non-malignant cells from 10 MF/SS patients (3 - 8 specimens per patient) representative of patients seen at the Washington University School of Medicine Cutaneous Lymphoma clinic (mean age 56.1 years, 50% female, 60% Caucasian and 40% African American). All were diagnosed with advanced MF and/or SS and half of first specimens were collected within 60 days of diagnosis. Subsequent specimens were collected at progression or lack of response to therapy, on average 20 months between specimens (1.4 - 60, SD = 11.8); patients had received 1-11 different CTCL therapies. For eWGS, specimens were purified by flow cytometry as follows, tumor: CD3+CD4+CD7- and normal: CD4+CD7+ (non-malignant T cells) or CD3-CD14+ (monocytes) or CD3-CD56+ (NK cells). Specimens were subjected to scRNAseq, exome sequencing plus low coverage whole genome sequencing (eWGS), and patient specific TCR clonotype assignment (scRepertoire).
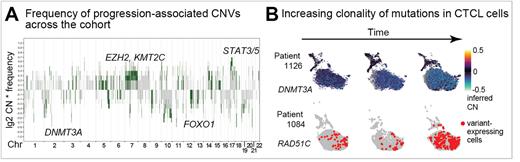
Results: Clustering and cell type inference of scRNAseq (UMAP/Seurat/singleR) from 92,496 total cells showed canonical CTCL marker genes and high clonal proportions (1-100%) of patient specific TCR clonotypes. Somatic genome copy number variants (CNV) from eWGS (cnvkit) and scRNAseq (copykat) and deleterious/damaging single nucleotide variants (SNV) (eWGS, GATK/Mutect2; scRNAseq, cb_sniffer) were identified in CTCL versus matched, non-malignant cells. To segregate early genomic events from those that were progression-associated, we developed an integrative analysis pipeline to prioritize somatic CNV and SNV that were detected in samples collected at timepoints with lack of response to therapy and/or progression in ≥30% of patients. This novel approach revealed alterations in T-cell activation pathways as early events that were maintained in subsequent CTCL samples. Loss of some chromatin-modifying enzymes were early events, while gains of others (DNMT3A, EZH2, KMT2C) were acquired with therapy resistance/progression (Fig. 1A). Copy number amplifications of STAT3/STAT5 are frequent and we demonstrate a novel-to-CTCL single nucleotide mutation in STAT3 that has gain-of-function activity. Progression-associated genomic changes were enriched in cell cycle (CDK genes), DNA damage/repair (TP53BP1, RAD51C), RAS and Rho signaling pathways, and transcriptional regulators (FOXO genes). Expression of mutant alleles was detectable in scRNAseq and variant levels fluctuated with progression or lack of therapy response (Fig. 1B).
Conclusion: To date, there have been limited studies defining temporal changes in the genomes and transcriptomes of CTCL due in part to low tumor burden in the skin and blood. Using innovative sequencing and informatic approaches, we identified recurrent alterations in CDK11A, DNMT3A, EZH2, FOXO1, RAD51C, STAT3/STAT5, and TP53BP1, among others, with clonal evolution in CTCL. Many of these have therapeutic implications. Taken together, these findings represent new insights into MF/SS progression, reveal new markers for response assessment, and identify new targets for therapeutic development.
Disclosures
Cooper:Indapta Therapeutics: Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Orca Biosystems: Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; American Association of Immunologists: Membership on an entity's Board of Directors or advisory committees; Affirmed: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Sobi: Consultancy; Miltenyi: Consultancy, Membership on an entity's Board of Directors or advisory committees; Wugen: Consultancy, Current holder of stock options in a privately-held company, Other: licensing; Immunity Bio: Research Funding; BMS: Research Funding; HCW Biologics: Research Funding. Musiek:Menlo: Research Funding; Pfizer: Research Funding; Connect: Research Funding; Elorac: Research Funding; UpToDate: Honoraria; AAD Board Prep Plus: Honoraria; Aristea: Research Funding; Bristol Myers Squib: Research Funding. Mehta-Shah:Genetech/Roch: Membership on an entity's Board of Directors or advisory committees, Research Funding; Daiichi Sankyo: Membership on an entity's Board of Directors or advisory committees, Research Funding; Corvus Pharmaceuticals: Research Funding; Celgene: Research Funding; Bristol Myers-Squibb: Research Funding; AstraZeneca: Consultancy, Membership on an entity's Board of Directors or advisory committees; Karyopharm Therapeutics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Kyowa Hakko Kirin Co., Ltd.: Membership on an entity's Board of Directors or advisory committees; Secura Bio: Membership on an entity's Board of Directors or advisory committees, Research Funding; Verastem: Research Funding. Payton:Merck: Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.