TO THE EDITOR:
Primary immunodeficiency diseases, including severe combined immunodeficiency (SCID) and combined immunodeficiency (CID), are monogenic disorders with a wide spectrum of clinical phenotypes including immune dysregulation, autoimmunity, inflammation, and malignancy.1 Manifestations may vary depending on whether the defect is localized to the immune system or its expression overlaps with other organ systems. Genetic aberrations affecting metabolic pathways, such as the coenzyme folate, can lead to immunodeficiency.2,3 SCID has previously been reported in patients with MTHFD1 deficiency, encoding a key enzyme in the folate pathway.2,4 Folates are essential for the synthesis of purine and pyrimidines during DNA and RNA synthesis and are cofactors for tissue growth and development.5,6 Folate uptake in humans is dependent on 2 transporters: the ubiquitously expressed reduced folate carrier (RFC) encoded by solute carrier family 19 member 1 (SLC19A1) and the gut epithelial proton-coupled folate transporter encoded by SLC46A1. Hereditary folate malabsorption caused by SLC46A1 mutations can present with SCID-like features,3,7,8 but immune defects due to SCL19A1 have never been reported. Indeed, a recent case described a deletion within SLC19A1 manifesting during teenage years with megaloblastic anemia but without immune abnormalities.9 We identify here, for the first time, 2 patients with primary immunodeficiency caused by a homozygous missense mutation in SLC19A1.
Both patients experienced severe recurrent infection, neurologic and hematologic disorders, and gastroenteropathy. Patient 1 (P1) is a 9-year-old boy. From infancy, he suffered oral lesions, recurrent fever, diarrhea, vomiting, atopic dermatitis, dark discoloration patches on his skin, and lip fissures (Figure 1A). He gradually developed curly gray hair and at 4 years of age suffered multiple deep abscesses on the buttocks and scalp. Owing to difficulty eating, he was fed a liquid diet until age 4 years and has multiple food allergies. He suffered chronic diarrhea and frequent respiratory infections requiring hospitalization. His growth and development were delayed, and magnetic resonance imaging of his brain showed extensive periventricular white matter hyperintensities (lesions) and undeveloped myelinization for his age (supplemental Figure 1A, available on the Blood website).
Patient clinical presentation and aberrant activation of stimulator of interferon genes (STING) and NF-κB pathway by G348R recombinant RFC (rRFC). (A) Oral lesions (i) and dark skin discoloration of the knee (ii) and arm (iii) of P1. (B) Immunologic characteristics prior to folate supplementation. Phytohemagglutinin stimulation index reflects values during supplementation. (C) Serum and red blood cell (RBC) folate levels in P1 and P2 during supplementation (ages 8 and 5 years, respectively) demonstrating low RBC folate levels (below the fifth percentile) despite normal serum levels. The solid line represents median and dotted lines represent 5th and 95th percentile concentrations of RBC folate in healthy controls.10 (D) Schematic diagram of the folate and CDN transporter RFC, showing 12 TMDs and the intracellular N-terminus and C-terminus. The G348R mutation affects the ninth transmembrane domain. (E) HEK293T cells expressing wild-type (WT) rRFC, G348R rRFC (G348R), or plasmid vector control (Vec) alongside STING were coincubated in the presence/absence of the cyclic dinucleotide 2’3’-cGAMP (10 μg/mL, 2 hours). Phosphorylation of STING (p-STING) was assessed by Western blotting. The G348R variant induced less STING phosphorylation. The G348R variant induced less STING phosphorylation (bottom), as quantified by densitometry (mean ± standard deviation of 4 independent experiments; 2-tailed Mann-Whitney test). (F) Induction of p65 phosphorylation (p-p65) was impaired following 2’3’-cGAMP incubation in G348R compared with WT rRFC expressing HEK293T cells, as quantified by densitometry (mean ± standard deviation of 3 independent experiments; 2-tailed Mann-Whitney test). (G) Downstream induction of IFN-β, assessed by quantitative polymerase chain reaction, revealed impaired messenger RNA (mRNA) gene expression in G348R expressing HEK293T cells compared with WT (replicates n = 3). (H) Incubation of patient lymphocytes (P1, P2) with 2’3’-cGAMP (40 μg/mL) resulted in reduced STING phosphorylation compared with the healthy controls (Ctrl). Results are normalized against total STING protein levels (bottom). IgG, immunoglobulin G; PHA, phytohemagglutinin; WBC, white blood cell.
Patient clinical presentation and aberrant activation of stimulator of interferon genes (STING) and NF-κB pathway by G348R recombinant RFC (rRFC). (A) Oral lesions (i) and dark skin discoloration of the knee (ii) and arm (iii) of P1. (B) Immunologic characteristics prior to folate supplementation. Phytohemagglutinin stimulation index reflects values during supplementation. (C) Serum and red blood cell (RBC) folate levels in P1 and P2 during supplementation (ages 8 and 5 years, respectively) demonstrating low RBC folate levels (below the fifth percentile) despite normal serum levels. The solid line represents median and dotted lines represent 5th and 95th percentile concentrations of RBC folate in healthy controls.10 (D) Schematic diagram of the folate and CDN transporter RFC, showing 12 TMDs and the intracellular N-terminus and C-terminus. The G348R mutation affects the ninth transmembrane domain. (E) HEK293T cells expressing wild-type (WT) rRFC, G348R rRFC (G348R), or plasmid vector control (Vec) alongside STING were coincubated in the presence/absence of the cyclic dinucleotide 2’3’-cGAMP (10 μg/mL, 2 hours). Phosphorylation of STING (p-STING) was assessed by Western blotting. The G348R variant induced less STING phosphorylation. The G348R variant induced less STING phosphorylation (bottom), as quantified by densitometry (mean ± standard deviation of 4 independent experiments; 2-tailed Mann-Whitney test). (F) Induction of p65 phosphorylation (p-p65) was impaired following 2’3’-cGAMP incubation in G348R compared with WT rRFC expressing HEK293T cells, as quantified by densitometry (mean ± standard deviation of 3 independent experiments; 2-tailed Mann-Whitney test). (G) Downstream induction of IFN-β, assessed by quantitative polymerase chain reaction, revealed impaired messenger RNA (mRNA) gene expression in G348R expressing HEK293T cells compared with WT (replicates n = 3). (H) Incubation of patient lymphocytes (P1, P2) with 2’3’-cGAMP (40 μg/mL) resulted in reduced STING phosphorylation compared with the healthy controls (Ctrl). Results are normalized against total STING protein levels (bottom). IgG, immunoglobulin G; PHA, phytohemagglutinin; WBC, white blood cell.
Patient 2 (P2) is a distant relative of P1, sharing a common ancestor 5 generations back. During infancy, he had repeated episodes of oral lesions, fever, respiratory infections, and chronic diarrhea necessitating multiple hospital admissions. Epilepsy and delayed development were also diagnosed. At 18 months, he was hospitalized for oral lesions, cracked lips, and severe bleeding from his mouth and nose and required thrombocyte transfusion owing to low platelet counts. By age 2 years, he developed genital dermatitis with abscess formation, dental caries, and gray sparse hair, which was previously black. Tomography scan of his brain revealed cerebral calcification at the bilateral caudate lobes, thalamus, basal ganglia, and right parietal and left frontotemporal lobes (supplemental Figure 1B). At 5 years of age, he could not speak and had difficulty walking.
Both patients had anemia (megaloblastic), thrombocytopenia, hypogammaglobulinemia, and lymphopenia (Figure 1B).
Whole exome sequencing on P1 identified only 1 variant predicted to have high impact and to potentially explain the patient’s phenotype, a homozygous missense chr21:46950793:C:T (hg19) SLC19A1 NM_194255:c.1042G>A (p.G348R), encoding RFC. Sanger sequencing and segregation analysis confirmed the presence of the homozygous SLC19A1 c.1042G>A (G348R) missense variant in P1, with a heterozygous state in his family (supplemental Figure 2A-B). The same homozygous SLC19A1 variant was identified in P2, whereas his healthy family members were heterozygous (supplemental Figure 2C-D).
RFC contains 12 transmembrane domains (TMDs) and localizes to the plasma membrane,11 where it facilitates transport of folate and cyclic dinucleotides (CDNs).12 G348R is located within the ninth TMD (Figure 1D), spanning residues 338 to 354,11 and displays a high level of conservation (supplemental Figure 3). Accordingly, missense impact prediction scores of MutationAssessor (3.28),13 SIFT (0.01),14 and PolyPhen2 HVAR (0.98)15 suggested this variant was deleterious. Modeling using TrRosetta predicted the G348R substitution to radically alter local protein structure and close-by transmembrane α-helices in the tertiary structure, interfering with transporter orientation or membrane insertion (supplemental Figure 2E).16 Red cell folate levels were greatly reduced in both patients despite normal serum folate levels (Figure 1C), indicating impairment of folate intake. Notably, confocal imaging of transfected cells showed that whereas G348R RFC was able to reach the plasma membrane (supplemental Figure 4A-B), its reduced expression in primary lymphocytes may indicate that it is less stable and likely targeted for degradation (supplemental Figure 4C).
In addition to transport of folate, RFC is a key transporter of CDNs. CDNs serve as “danger signals,” indicating the presence of foreign bacteria or tumorigenic cells. RFC facilitates movement of these charged immunoreactive molecules across the plasma membrane to activate the STING pathway and downstream signaling. Despite G348R membrane expression in transfected HEK293 cells, phosphorylation of STING was reduced following stimulation with 2’3’-cyclic guanosine monophosphate–adenosine monophosphate (cGAMP) (Figure 1E). Downstream NF-κB activation and induction of interferon-β (IFN-β) transcription, both essential components of the STING-mediated response, were also significantly reduced (Figure 1F-G). In agreement, cGAMP-induced STING phosphorylation was impaired in patient lymphocytes (Figure 1H). Thus, the G348R RFC mutation showed a diminished capacity to facilitate cGAMP intracellular signaling and gene induction, consistent with significantly reduced transporter activity. CDN 2’3’-cGAMP and 3’3’-cGAMP are potent adjuvants, normally driving expansion and maturation of antigen-specific CD8+ T cells via the induction of type I IFNs, which also activate innate immune responses.17-19
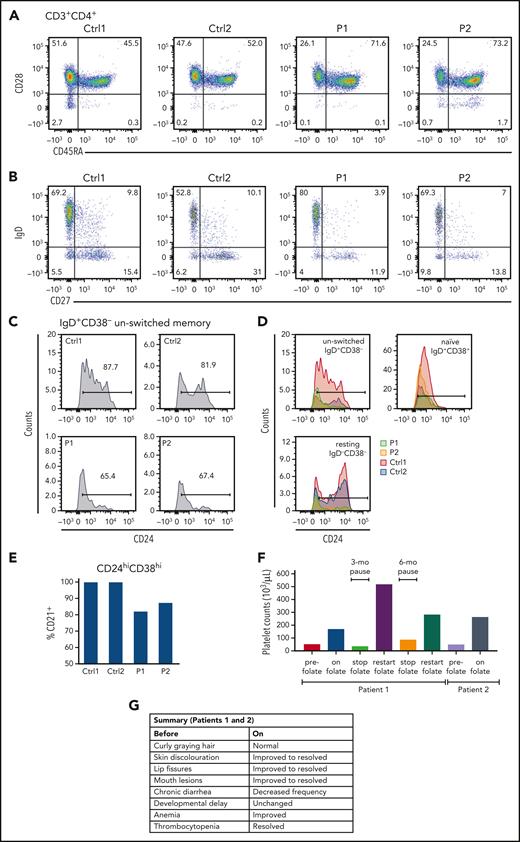
RFC transport of folate is critical to nucleic acid synthesis and formation of DNA, both impacting cell division, with defects likely preferentially affecting immune cells such as lymphocytes that undergo rapid clonal expansion. Both patients had low CD4+ T cells, and in vitro responses to PHA were depressed (Figure 1B). An abnormally increased number of terminally differentiated (CD45RA+CD28−) CD4 T cells was present in P2 (Figure 2A), and there were increased levels of CD57 on memory cells, potentially reflecting defective proliferative and functional capacity, in both patients (Ctrl1 [C1] and Ctrl2 [C2], 9.25% and 8.05%, respectively; P1 and P2, 13.9% and 15.3%, respectively; supplemental Figure 5A). Furthermore, follicular helper T cells (CXCR5+) were low (C1 and C2, 19% and 22%, respectively; P1 and P2, 10.5% and 5.9%, respectively; supplemental Figure 5B).
Lymphocyte subpopulation analysis and improvement in SLC19A1-deficient patients following folic supplementation. (A) Analysis of T cell populations revealed mature CD4+ cells in P1 and P2, with an unusually high number of terminally differentiated CD4 T cells present in P2. (B) Analysis of B cell differentiation confirmed the presence of class-switched IgD-CD27+ cells in both patients (P1, P2) as well as mature activated IgD+CD27+ B cells, albeit at lower levels in P1. Results from 2 controls (Ctrl1, Ctrl2), both healthy, are shown for comparison. (C-D) P1 and P2 exhibited reduced subpopulations of CD24hi unswitched memory cells compared with controls. (E) Normal levels of CD24hi transitional B lymphocytes were identified in both patients, although 10% to 20% of these cells failed to express CD21. (F) Platelet counts before and during (on) folic acid supplementation and during periods when treatment was paused. (G) Summary of improvements observed in P1 and P2 before and during (on) supplementation.
Lymphocyte subpopulation analysis and improvement in SLC19A1-deficient patients following folic supplementation. (A) Analysis of T cell populations revealed mature CD4+ cells in P1 and P2, with an unusually high number of terminally differentiated CD4 T cells present in P2. (B) Analysis of B cell differentiation confirmed the presence of class-switched IgD-CD27+ cells in both patients (P1, P2) as well as mature activated IgD+CD27+ B cells, albeit at lower levels in P1. Results from 2 controls (Ctrl1, Ctrl2), both healthy, are shown for comparison. (C-D) P1 and P2 exhibited reduced subpopulations of CD24hi unswitched memory cells compared with controls. (E) Normal levels of CD24hi transitional B lymphocytes were identified in both patients, although 10% to 20% of these cells failed to express CD21. (F) Platelet counts before and during (on) folic acid supplementation and during periods when treatment was paused. (G) Summary of improvements observed in P1 and P2 before and during (on) supplementation.
Circulating CD19+ B cells were borderline low, with low serum IgG requiring replacement with IV immunoglobulin. Typical B cell subsets were present, but CD24hi cells were selectively reduced in resting and unswitched memory cells. CD24 expression upregulates in CD27+ memory cells, decreases during class-switching, and returns to low expression in plasmablasts, suggesting either CD24 induction is aberrant or shedding of the glycosylphosphatidylinositol-anchored protein is increased (Figure 2B-D). Approximately 20% of CD24hiCD27+ B cells express interleukin-10 (IL-10) and can act as suppressors of CD4+ T cell function.20,21 Furthermore, analysis of CD38hiCD24hi transitional B cells were found within normal range (Figure 2C-D), but CD21 expression by these cells was reduced (Figure 2E). CD21+ transitional B cells contain IL-10–producing cells in the mouse.20 If CD21+ transitional B cells are similar in humans, and if the reduction in CD24hi cells include a concomitant decline in IL-10–secreting cells, it could be hypothesized that there might be a defect of B cell regulation in these patients.
The ability of folate supplementation to resolve many clinical and laboratory features supported the pathogenicity of the G348R RFC variant. Folic acid supplementation (2.5 mg per day) was started in P1 at 5.5 years of age and at 3.5 years in P2. Platelet counts normalized rapidly, and anemia improved (Figure 2F, supplemental Figure 8). Hair and skin discoloration, mouth lesions, and chronic diarrhea improved in both patients. In contrast, neurologic and developmental issues remained unchanged, and treatment had only a partial effect on the immune system (Figure 2G). CD4+ counts normalized, but mitogen responses and circulating B cell numbers remained depressed. Clinical response over time in P2 was difficult to assess as supplementation was inconsistent. Growth and development were severely delayed, and at 6.5 years of age, he developed acute strabismus and was unable to walk or recognize family members, dying 7 days later. Potentially, high-does folate supplementation in early pregnancy might be considered to limit neurologic damage in future cases with prior genetic diagnoses.21
In sum, we have shown for the first time that mutations in RFC can be associated with combined immunodeficiency; hematologic abnormalities; and skin, hair, and neurologic disorders (supplemental Figure 9), which are partially reversible by folic acid supplementation.
Acknowledgments
This work was supported by Immunodeficiency Canada’s Distinguished Professorship in Immunology (C.M.R.), the Program for Immunogenomics and the Canadian Centre for Primary Immunodeficiency (C.M.R.), the Jeffrey Modell Foundation, and Immunodeficiency Canada (C.M.R.). A.S. is supported by the Department of Pediatrics, Graduate School of Medicinal Sciences, Kyushu University, Japan; the International Research Fund for Subsidy of Kyushu University School of Medicine Alumni, Japan; and the CAAIF/Immunodeficiency Canada Research Fellow/Early Investigator Award in Immunodeficiency.
Authorship
Contribution: C.M.R. performed the study conception; A.S., C.M.R., N.S., L.V., and S.G. designed the study; R.A., V.U., N.E.K., S.M., and C.M.R. performed clinical evaluation; A.S., N.S., H.D., S.B., and M.M. performed laboratory and data analysis; D.M. performed exome variant prioritization analysis; M.G.F.S. performed structural modeling analysis; A.S., C.M.R., N.S., L.V., D.M., and M.G.F.S. wrote the first manuscript draft; and all authors critically revised and approved the final version of the article.
Conflict-of-interest disclosure: D.M. was a full-time employee and is currently a shareholder of Deep Genomics. M.G.F.S. was a full-time employee of Deep Genomics and entitled to a stock option during this work. The remaining authors declare no competing financial interests.
Correspondence: Chaim M. Roifman, Division of Immunology and Allergy, Hospital for Sick Children, 555 University Ave, Toronto, ON M5G 1X8, Canada; e-mail: chaim.roifman@sickkids.ca.
References
Author notes
Complete whole exome sequencing data are available upon request from the corresponding author, Chaim M. Roifman (chaim.roifman@sickkids.ca).
The online version of this article contains a data supplement.