Key Points
Cilta-cel, a BCMA-targeting chimeric antigen receptor−T-cell therapy, induced clinical responses in patients previously exposed to noncellular anti-BCMA therapies.
Results of this study may inform sequencing strategies for anti-BCMA therapies in patients with relapsed/refractory MM.
Abstract
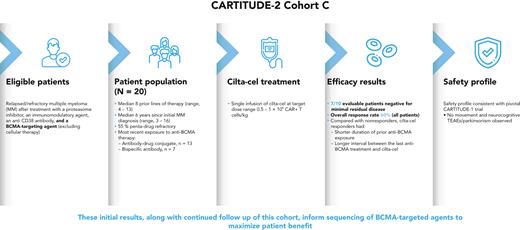
B-cell maturation antigen (BCMA)–targeting therapies, including bispecific antibodies (BsAbs) and antibody-drug conjugates (ADCs), are promising treatments for multiple myeloma (MM), but disease may progress after their use. CARTITUDE-2 is a phase 2, multicohort study evaluating the safety and efficacy of cilta-cel, an anti-BCMA chimeric antigen receptor T therapy, in various myeloma patient populations. Patients in cohort C progressed despite treatment with a proteasome inhibitor, immunomodulatory drug, anti-CD38 antibody, and noncellular anti-BCMA immunotherapy. A single cilta-cel infusion was given after lymphodepletion. The primary end point was minimal residual disease (MRD) negativity at 10−5. Overall, 20 patients were treated (13 ADC exposed; 7 BsAb exposed; 1 in the ADC group also had prior BsAb exposure). Sixteen (80%) were refractory to prior anti-BCMA therapy. At a median follow-up of 11.3 months (range, 0.6-16.0), 7 of 20 (35%) patients were MRD negative (7 of 10 [70.0%] in the MRD-evaluable subset). Overall response rate (95% confidence interval [CI]) was 60.0% (36.1-80.9). Median duration of response and progression-free survival (95% CI) were 11.5 (7.9—not estimable) and 9.1 (1.5—not estimable) months, respectively. The most common adverse events were hematologic. Cytokine release syndrome occurred in 12 (60%) patients (all grade 1-2); 4 had immune effector cell-associated neurotoxicity syndrome (2 had grade 3-4); none had parkinsonism. Seven (35%) patients died (3 of progressive disease, 4 of adverse events [1 treatment related, 3 unrelated]). Cilta-cel induced favorable responses in patients with relapsed/refractory MM and prior exposure to anti-BCMA treatment who had exhausted other therapies. This trial was registered at www.clinicaltrials.gov as NCT04133636.
Introduction
B-cell maturation antigen (BCMA), expressed predominantly on the surface of plasma cells, is a promising target for antimyeloma therapy.1 Several treatments specifically targeting BCMA-expressing cells are under investigation or approved for the treatment of multiple myeloma (MM), including bispecific antibodies (BsAbs), antibody-drug conjugates (ADCs), and chimeric antigen receptor (CAR)−T-cell therapies.2-5 Recent studies of anti-BCMA BsAbs, including elranatamab, teclistamab, AMG420, Tnb-383B (ABBV-383), CC-93269 (alnuctamab), and REGN5458, as well as the anti-BCMA ADC, belantamab mafodotin, demonstrate that patients may experience disease progression after treatment, leaving them with few other treatment options.6-12
Ciltacabtagene autoleucel (cilta-cel) is a CAR−T-cell therapy expressing 2 BCMA-targeting single-domain antibodies designed to confer avidity, a CD3-ζ signaling domain, and a 4-1BB costimulatory domain to optimize T-cell activation and proliferation.13 Initial results from the phase 1b/2, single-arm CARTITUDE-1 trial demonstrated that cilta-cel led to early, deep, and durable responses in heavily pretreated patients with relapsed/refractory MM (RRMM).13 At a median follow-up of ∼2 years, overall response rate (ORR) was 98% (stringent complete response [sCR], 83%). Median duration of response (DOR) and median progression-free survival (PFS) were not reached.14
CARTITUDE-1, along with most clinical trials for BCMA-targeting therapies in MM, excluded patients who have previously received BCMA-directed therapy.13,15 However, there have been reported cases of individuals who progressed after an initial anti-BCMA therapy, maintained BCMA expression, and responded to a different anti-BCMA therapy.16 Thus, there is a need to identify the optimal sequencing of BCMA-directed agents in MM. CARTITUDE-2 is a phase 2, multicohort, open-label study evaluating cilta-cel safety and efficacy in several patient populations. Here, we report initial results from cohort C, which enrolled quadruple-class exposed patients with RRMM who had prior exposure to noncellular BCMA-directed agents.
Methods
Study design and treatment
CARTITUDE-2 is an ongoing, phase 2, multicohort, open-label, multicenter study of cilta-cel (ClinicalTrials.gov identifier: NCT04133636). Multiple patient populations with unmet need are being evaluated, with approximately 20 to 40 patients in each cohort. Cohort C is evaluating cilta-cel in patients with heavily pretreated RRMM, who were previously exposed to BCMA-targeted therapy (excluding cellular immunotherapy).
Eligible patients were ≥18 years of age, had a diagnosis of MM per International Myeloma Working Group (IMWG) diagnostic criteria17 and evidence of progressive disease (PD) within 12 months of their last line of therapy (LOT), or within 6 months of prior therapy and refractory to their most recent LOT, had measurable disease at baseline and an European Cooperative Oncology Group performance status score of 0 or 1. Patients must have been previously treated with a proteasome inhibitor, an immunomodulatory drug, an anti-CD38 mAb, and a noncellular BCMA-directed therapy (ADC or BsAb as monotherapy or combination LOT). Patients were eligible regardless of dose level/schedule or response to prior BCMA-directed therapy. BCMA expression was not required for eligibility.
Patients received a single cilta-cel infusion (target dose 0.75 × 106 CAR-positive viable T cells/kg; target range, 0.5-1.0 × 106) 5 to 7 days after initiation of lymphodepletion (300 mg/m2 cyclophosphamide and 30 mg/m2 fludarabine daily for 3 days). Retreatment with cilta-cel was permitted within the same dose range in patients with documented disease progression ≥6 months after cilta-cel infusion, with at least minimal response, who had no ongoing hematologic (grade ≥3) or nonhematologic (grade ≥2) toxicities.
An independent ethics committee or institutional review board at each study center approved the study protocol, and all patients provided written informed consent. A data monitoring committee provided oversight and recommendations for interventions to improve safety. The study protocol was conducted in accordance with the Declaration of Helsinki and International Conference on Harmonisation guidelines for Good Clinical Practice.
End points and assessments
The primary end point was minimal residual disease (MRD) negativity at 10−5 as defined by the IMWG criteria,17-19 assessed centrally by next-generation sequencing (clonoSEQ v2.0; Adaptive Biotechnologies, Seattle, WA). MRD was assessed at day 56 and at 6, 12, 18, and 24 months regardless of disease status; an additional sample was collected and assessed at the time of suspected complete response (CR) and then every 12 months until disease progression for patients who remained on study. MRD negativity was assessed in samples that passed calibration or quality control and included sufficient cells for evaluation at the testing threshold of 10−5. A patient was considered to have achieved MRD negativity if ≥1 postbaseline assessment was negative. CR was not a criterion for the primary end point.
Secondary end points were incidence and severity of adverse events (AEs), ORR, rates of very good partial response (VGPR), CR, and sCR based on IMWG response criteria,17-19 as well as DOR and time to response. PFS was an exploratory end point.
AEs were graded using National Cancer Institute Common Terminology Criteria for Adverse Events (NCI-CTCAE) v5.0. Cytokine release syndrome (CRS) and immune effector cell–associated neurotoxicity syndrome (ICANS) were graded according to American Society for Transplantation and Cellular Therapy criteria.20 Other neurotoxicities (events not reported as ICANS) were graded using NCI-CTCAE v5.0.
Serum/urine disease evaluation was performed at screening; during conditioning; on days 28, 56, 78, 100, and every 28 days up to 12 months; and then every 56 days. Immunoglobulins were measured at screening; at baseline (before cilta-cel infusion); on days 28, 56, 78, and 100; and then every 28 days up to 12 months, followed by every 56 days. Anti-drug antibodies (ADAs) were measured on day 1 predose; on days 14, 28, 56, 78, 100, and 184; and at PD or study completion).
CAR transgene levels in blood and soluble serum BCMA were analyzed at baseline; on day 1 predose and post cilta-cel infusion; on days 2, 3, 7, 10, 14, 21, 28, 42, 56, 78, and 100; and then every 8 weeks up to 1 year. A similar schedule of analysis was used for immunophenotyping (plus a reading at 35 days) and cytokine profiling (last reading at 100 days).
Statistical analysis
The sample size of this cohort was selected to collect preliminary data on efficacy and safety. For the primary end point, the MRD-negative rate was defined as the proportion of patients negative for MRD by bone marrow aspirate at any time point after the initial dose of cilta-cel and before disease progression or starting subsequent therapy including retreatment with cilta-cel. The MRD-negative rate and its 2-sided 95% Clopper-Pearson exact CI were determined in MRD-evaluable patients and in all treated patients.
ORR was defined as the proportion of patients who achieve at least partial response according to the IMWG criteria.17-19 Response to treatment was analyzed by a validated computerized algorithm.21,22 For patients with neither serum nor urine measurable disease and using positron-emission/computed tomography or whole-body magnetic resonance imaging to satisfy the measurable disease criteria per protocol, the response was based on the investigator’s assessment. ORR and its 95% CI were calculated by the Clopper-Pearson method. Time-to-event end points were assessed using Kaplan-Meier estimates. Time to response was summarized descriptively.
All authors had access to the primary clinical trial data; statistical analyses were performed by the study sponsor.
Results
Patients and treatment
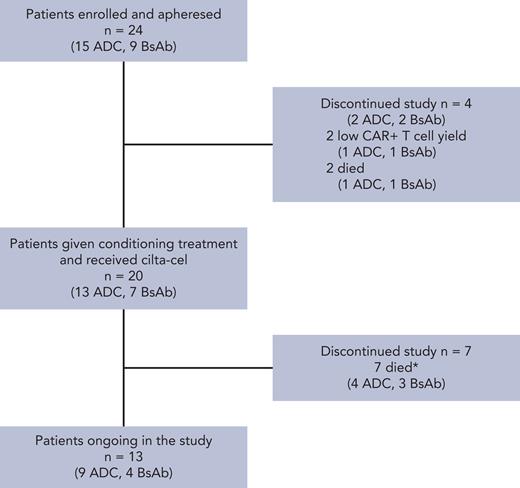
As of the 8 October 2021 data cutoff, 24 patients were enrolled and apheresed and 20 were treated with cilta-cel. Four patients did not receive cilta-cel because of either low cellular yield (n = 2; 1 patient with prior ADC and 1 with prior BsAb exposure) or death due to progressive disease before dosing (n = 2; 1 patient in each group). The absolute lymphocyte count inclusion requirement of ≥0.3 × 109/L was met for all patients at the time of screening and apheresis for all patients enrolled. The most recent anti-BCMA treatments were BsAb (n = 7 patients [35%]) and ADC (n = 13 [65%]). One patient received 2 different ADCs, and 1 patient received both ADC and BsAb (counted in the ADC-exposed group as the most recent exposure was to ADC). Six patients (30%) had received anti-BCMA treatment as their last LOT (n = 4 ADC, n = 2 BsAb; Table 1; supplemental Table 1, available on the Blood website). Duration of prior anti-BCMA treatment ranged from 1 to 527 days, time from last anti-BCMA agent to apheresis ranged from 26 to 895 days, and time from last anti-BCMA agent to cilta-cel infusion ranged from 62 to 944 days (Table 2). In the ADC group, 2 patients had a best response to anti-BCMA treatment of VGPR and 1 of sCR, whereas 1 patient each in the BsAb group had reached VGPR and CR; the rest of the patients had best response of SD or PD (1 patient was not evaluable [NE]; supplemental Table 1). Median PFS (95% CI) after treatment with the last prior anti-BCMA treatment was 1.9 (1.1-5.1) months in all patients, 1.8 (1.1-2.1) months in the ADC group, and 4.2 (0.3-6.7) months in the BsAb group. Patients discontinued their last prior anti-BCMA therapy due to PD (n = 15), AEs (n = 3), or both (n = 2). At baseline, median age for cohort C was 63 years (range, 44-81), 12 (60%) patients were male, and 19 (95%) were white and 1 (5%) was black. Five patients (25%) had baseline extramedullary disease, and 3 (15%) had high-risk cytogenetics at screening. Patients had received a median of 8 prior LOTs (range, 4-13); 95% were refractory to their last LOT, with 18 (90%) triple-class refractory, 11 (55%) penta-drug refractory, and 16 (80%) refractory to noncellular anti-BCMA treatment (Table 1).
Bridging therapies were variable, and 18 of 20 patients received bridging therapy before cilta-cel infusion (supplemental Table 2). Of the 2 patients who did not receive bridging therapy, 1 had increased tumor burden and 1 was NE. Of the patients who received bridging therapy, 6 had decreased tumor burden between screening and cilta-cel infusion, 3 were NE, 8 had increased tumor burden, and 1 had no change. Categorically, no patient achieved CR after bridging therapy. Median administered cilta-cel dose was 0.65 × 106 (range, 0.21 × 106 to 1.11 × 106) CAR-positive viable T cells/kg; 1 patient received a below-target dose.
Efficacy
Overall cohort
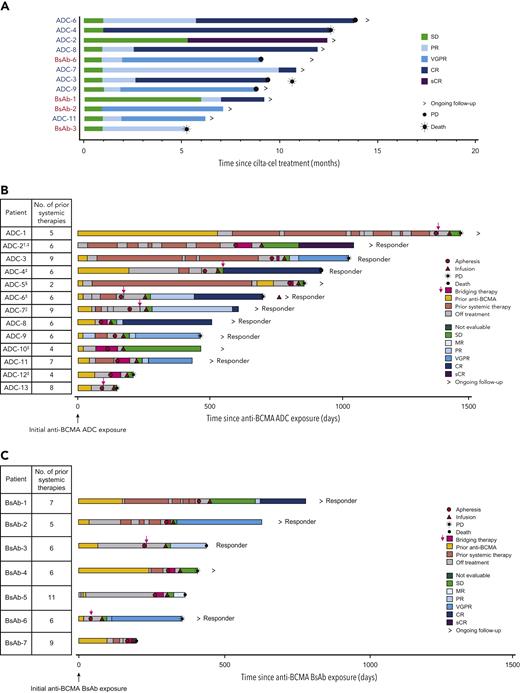
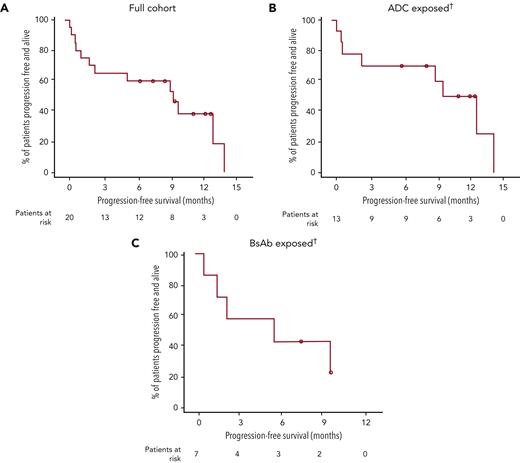
At the time of data cutoff, 13 of 20 patients (9 in the ADC group, 4 in the BsAb group) remained on study (Figure 1). At a median follow-up of 11.3 months (range, 0.6-16.0), 7 of 20 patients were MRD negative at any time at 10−5 (35%; 95% CI, 15.4-59.2); considering only the 10 patients who had MRD-evaluable samples, the proportion was 70.0% (95% CI, 34.8-93.3). The ORR for all patients in cohort C (n = 20) was 60.0% (95% CI, 36.1-80.9), with 55% of patients achieving ≥VGPR and 30% achieving ≥CR (Table 3). Of the 7 patients who achieved MRD negativity, clinical responses were sCR (n = 1), CR (n = 2), VGPR (n = 3), and PD (n = 1; due to increased plasmacytoma size). All 3 patients with high-risk cytogenetics (all del17p) achieved ≥VGPR (supplemental Table 1). The median time to first response was 0.95 months (range, 0.9-6.0), and median time to best response was 2.22 months (range, 0.9-9.9; Table 3; Figure 2A). Median DOR was 11.5 months (95% CI, 7.9-NE). Median PFS was 9.1 months (95% CI, 1.5-NE), and the 12-month PFS rate was 38.9% (95% CI, 16.3-61.1; Figure 3A). Median overall survival (OS) was not reached at data cutoff. In 4 of 5 patients with plasmacytoma at baseline, plasmacytomas decreased 100% after cilta-cel treatment.
Patient disposition. ∗Three were due to Parkinson disease, 2 to acute respiratory failure (COVID-19), 1 to subarachnoid hemorrhage (not treatment related), and 1 to Clostridium difficile colitis (treatment related).
Patient disposition. ∗Three were due to Parkinson disease, 2 to acute respiratory failure (COVID-19), 1 to subarachnoid hemorrhage (not treatment related), and 1 to Clostridium difficile colitis (treatment related).
Response to cilta-cel. Response among responders (A) and in all patients with previous exposure to antibody-drug conjugate (ADC) (B) or BsAb (C). Classification is based on the last anti−B-cell maturation antigen therapy used if patients received >1 therapy. NE, not evaluable; SD, stable disease. †Patient was treated with ADC for 1 day. ‡Patients with extramedullary plasmacytomas. §Patient was treated with 2 different ADCs. ||Patient received BsAb first and ADC later.
Response to cilta-cel. Response among responders (A) and in all patients with previous exposure to antibody-drug conjugate (ADC) (B) or BsAb (C). Classification is based on the last anti−B-cell maturation antigen therapy used if patients received >1 therapy. NE, not evaluable; SD, stable disease. †Patient was treated with ADC for 1 day. ‡Patients with extramedullary plasmacytomas. §Patient was treated with 2 different ADCs. ||Patient received BsAb first and ADC later.
Progression-free survival. Kaplan-Meier curves showing progression-free survival in the overall cohort (A), the antibody-drug conjugate (ADC)-exposed patients (B), and the BsAb-exposed patients (C). †Classification is based on the last anti-BCMA therapy used if patients received >1 therapy.
Progression-free survival. Kaplan-Meier curves showing progression-free survival in the overall cohort (A), the antibody-drug conjugate (ADC)-exposed patients (B), and the BsAb-exposed patients (C). †Classification is based on the last anti-BCMA therapy used if patients received >1 therapy.
Anti-BCMA ADC-exposed group
Patients who had prior exposure to ADC had a median follow-up of 11.8 months (range, 2.0-16.0) after cilta-cel infusion. Five patients achieved MRD negativity, for a rate of 39% (95% CI 13.9-68.4) considering all 13 ADC-exposed patients and 71.4% (95% CI, 29.0-96.3) considering the 7 MRD-evaluable patients. ORR was 61.5% (95% CI, 31.6-86.1), with median time to first response of 0.97 months (range, 0.9-5.1) and median time to best response of 2.58 months (range, 0.9-9.9; Table 3; Figure 2B). The 5 MRD-negative patients achieved best responses of sCR (n = 1), CR (n = 1), VGPR (n = 2), and PD (n = 1). Median DOR was 11.5 months (95% CI, 7.9-NE), and median PFS was 9.5 months (95% CI, 0.99-NE; Figure 3B. One of the 4 patients who received ADC as the last LOT responded to cilta-cel, whereas 7 of the 9 patients who received ≥1 other treatment between ADC and cilta-cel achieved a response (supplemental Table 1).
One patient in the ADC group was retreated with cilta-cel ∼16 months after initial infusion (doses of 0.58 and 0.53 × 106 CAR-positive viable T cells/kg). This patient achieved CR after the first infusion but had PD 8 months later. There were no therapies administered between cilta-cel infusions. The second infusion was given 4 days before the current data cutoff date; thus, response is not known at present.
Anti-BCMA BsAb-exposed group
Patients with prior BsAb exposure had a median follow-up of 10.9 months (range, 0.6-11.5) after cilta-cel infusion. Two of 7 patients achieved MRD negativity (2 of 3 considering only the evaluable patients). The 2 MRD-negative patients achieved CR and VGPR. ORR was 57.1% (95% CI, 18.4-90.1), with median time to first response of 0.92 months (range, 0.9-6.0) and median time to best response of 1.4 months (range, 0.9-7.0; Table 3; Figure 2C). Median DOR was 8.2 months (95% CI, 4.4-NE), and median PFS was 5.3 months (95% CI, 0.6-NE; Figure 3C). Both patients who received BsAb as their last LOT responded to cilta-cel. For patients who had another treatment between BsAb and cilta-cel, 2 of 5 responded (supplemental Table 1).
Factors associated with response
Several trends emerged in descriptive analyses of cilta-cel responders vs nonresponders. Patients who responded had shorter median duration of exposure to prior anti-BCMA treatment (29.5 days; range, 1-277) than those who did not respond (63.5 days; range, 22-527; Table 2). Most patients who responded to cilta-cel did not respond to their prior anti-BCMA therapy (10 of 12 cilta-cel responders did not respond to prior anti-BCMA therapy; supplemental Table 1). Responders to cilta-cel had a longer median time between the last anti-BCMA treatment and apheresis (161.0 days; range, 26-695) than nonresponders (56.5 days; range, 40-895) and a longer median time between the last anti-BCMA treatment and cilta-cel infusion (235.0 days; range, 62-749) than nonresponders (117.5 days; range, 95-944; Table 2).
Safety
All 20 patients experienced treatment-emergent adverse events (TEAEs) after treatment with cilta-cel, 19 of whom had TEAEs related to cilta-cel treatment; the worst toxicity grade of AEs was grade 2 (n = 1 patient), grade 3/4 (n = 17), and grade 5 (n = 2). Grade 3 to 4 AEs occurring in >1 patient were mainly cytopenias (neutropenia, n = 17; thrombocytopenia, n = 14; anemia, n = 11; leukopenia, n = 11; lymphopenia, n = 6) but also included hypocalcemia (n = 2), hypophosphatemia (n = 4), hypokalemia (n = 2), and ICANS (n = 2) (Table 4). On the basis of laboratory data, patients with grades 3 to 4 cytopenia events after cilta-cel infusion recovered to grade ≤2 by day 30 for neutropenia (79%), lymphopenia (70%), and thrombocytopenia (43%).
Overall, 12 (60%) patients had CRS (all grades 1-2) with a median time to onset of 7.5 days (range, 2-10) and median duration of 7 days (range, 2-9). All patients who had CRS received ≥1 treatment for it (9 tocilizumab, 3 anakinra, 6 corticosteroids); CRS resolved in all patients. Four patients had ICANS. Median time to onset was 9.0 days (range, 4-13), and median duration was 7.0 days (range, 4-20); 3 cases resolved. No other neurotoxicities, including movement and neurocognitive TEAEs/parkinsonism, were observed in this cohort.
Seven (35%) patients died after cilta-cel treatment. In the ADC group, there were 4 deaths: 3 due to PD and 1 due to COVID-19 pneumonia (not treatment related per investigator assessment). In the BsAb group, there were 3 deaths, all due to AEs (COVID-19 pneumonia [not treatment related per investigator assessment], Clostridium difficile colitis [treatment related], and subarachnoid hemorrhage [not treatment related]; supplemental Table 1). Of the 3 patients who died of infection, only 1 (prior ADC group) had received intravenous immunoglobulin (IVIG); altogether, 5 patients in the full cohort received IVIG. Median levels of immunoglobulins A and M in the full cohort were stable (0.28 and 0.20 g/L, respectively) from baseline to the most recent assessment (day 408).
Pharmacokinetics and biomarkers
Cilta-cel pharmacokinetics was characterized by CAR transgene levels and CAR-positive cells in peripheral blood and bone marrow. Following infusion there was an initial expansion phase of the CAR transgene, with time to maximum concentration around 15 days (range, 9-41 days), followed by a rapid decline phase and then a slower decline over months. The median time of last measurable CAR transgene was around 127 days (range, 15-213). High interindividual variability was observed (supplemental Figure 1). CAR transgene exposure parameters including maximum concentration (Cmax) and area under the concentration-time curve from time 0 to 28 days (AUC0-28d) showed slightly higher mean values in the BsAb group than in the ADC group (supplemental Figure 2). Similar patterns were observed when measuring CAR-positive cells in blood.
At apheresis, the BsAb group had a higher frequency of T-effector memory cells, especially in the CD8 compartment, than the ADC group. Consistent with a more activated phenotype, these patients had higher postbaseline peak median levels of interleukin (IL)-6, interferon-γ, IL-2, and soluble IL-2 receptor-α vs the ADC group. Baseline IL-10 levels were elevated in the BsAb group compared with the ADC group.
Before cilta-cel treatment, the mean serum BCMA baseline level was 211 µg/L (range, 0.4-943) and 203 µg/L (range, 1.3-541) for the ADC and BsAb groups, respectively. Serum BCMA decreased after cilta-cel infusion, with mean serum concentrations reaching nadir levels near the lower limit of quantification around day 100 (supplemental Figure 3). Of 9 patients with PD on study (7 from ADC group and 2 from BsAb group), all had serum BCMA levels at the time of progression similar to baseline before cilta-cel treatment. No correlation was identified between baseline serum BCMA and clinical response.
Three patients in the ADC group (23%) were positive for ADAs at baseline, and there was no increase in ADA observed during follow-up; no patients in the BsAb group were positive at baseline or during follow-up.
Discussion
In cohort C of CARTITUDE-2, quadruple-class exposed patients with RRMM who had previously received a noncellular BCMA-targeting treatment had an MRD negativity rate of 70% in evaluable patients (35% in the full cohort). The ORR to cilta-cel at median 11.3 months of follow-up was 60%, with median DOR of 11.5 months and median PFS of 9.1 months, indicating that sequencing cilta-cel after prior BCMA-targeted therapy can offer a meaningful clinical benefit to patients who have few other options. The cohort C population comprised patients with aggressive, refractory disease (median 8 prior LOTs and 80% refractory to prior anti-BCMA). This was reflected in the number of patients who died before cilta-cel infusion or who had insufficient apheresis material to manufacture cilta-cel. The proportion of patients who underwent apheresis but not CAR-T cell infusion was similar to that of CARTITUDE-1. These data argue for earlier referral for cilta-cel treatment to increase successful infusion.
Although the response rate in CARTITUDE-2 cohort C was lower than that of CARTITUDE-1 patients, cilta-cel efficacy is still favorable compared with other myeloma treatments administered after anti-BCMA therapy.13 Recent data from the STOMP phase 1b/2 trial of selinexor combination treatments in patients previously treated with an anti-BCMA therapy showed an ORR of 55%, with a median OS of 10.5 months and a 6-month PFS rate of 69%.23 In addition, patients with prior anti-BCMA treatment who were treated with cevostamab (anti-FcRH5 × CD3) monotherapy had ORRs ranging from 33.3% to 50%, depending on the dose of cevostamab and prior treatment type.6,24 Patients treated with iberdomide and dexamethasone after anti-BCMA treatment had an ORR of 25%.25 In addition, 3 of 4 patients previously exposed to anti-BCMA treatments responded to elranatamab.26
CARTITUDE-1 established that patients who are naïve to anti-BCMA therapy clearly benefit from cilta-cel treatment, with a response rate of 98% and median DOR and median PFS not reached at 27.7 months’ follow-up vs other approved anti-BCMA therapies (ide-cel, ORR 73%; belantamab mafodotin, ORR 31%).14,15,27 In patients already exposed to BCMA-targeting therapies, the CARTITUDE-2 cohort C results suggest that a large proportion (60%) may still respond to cilta-cel. Although the CARTITUDE-1 data support using cilta-cel in anti-BCMA naïve patients, which appears to lead to better clinical response rates, access limitations to CAR-T cell therapeutics may necessitate use of other BCMA-targeting therapies before cilta-cel. Appropriate patient selection and prior treatment sequencing may also increase the response rate. Factors associated with response to cilta-cel were (1) a shorter duration of the last anti-BCMA treatment and (2) a longer interval between anti-BCMA treatment and cilta-cel. The latter could possibly be related to intervening therapies and/or potentially less aggressive disease in cilta-cel responders. These findings need to be validated in larger cohorts but suggest that these factors should be taken into consideration when determining candidacy for cilta-cel treatment after other BCMA-targeting therapies. Whether noncellular BCMA-targeting therapy can be used as bridging therapy before BCMA-targeting CAR-T cell therapy and whether patients who respond to prior BCMA-targeting therapy will have better outcomes when sequencing multiple BCMA-targeting therapeutics remain unknown.
The safety profile of cilta-cel in cohort C of CARTITUDE-2 was consistent with that of CARTITUDE-1.13 The frequency of grade 3/4 cytopenias, as well as recovery time from these, were consistent with CARTITUDE-1. The benefits of cilta-cel in this population appeared to outweigh the risks, with no new safety signals observed. In CARTITUDE-1, 6% of patients exhibited signs and symptoms of movement and neurocognitive TEAEs/parkinsonism, but strategies to manage these TEAEs were implemented in CARTITUDE-2 and across the rest of the clinical development program, including more effective bridging therapy, early and aggressive treatment of CRS and ICANS, handwriting assessment, and extensive monitoring.28 Apart from ICANS, no other neurotoxicities were observed in cohort C. The 3 infection-related deaths in this cohort reflect the profound immune suppression experienced by late-stage myeloma patients, which may be exacerbated by BCMA-targeted therapies, highlighting the need for close monitoring for infectious symptoms and consideration for preventive measures such as IVIG and/or SARS-CoV2-specific monoclonal antibodies, if indicated.29
In general, pharmacokinetic measurements using both transgene and cellular levels were concordant and showed similar profiles regardless of prior ADC or BsAb exposure. CAR-T cell expansion kinetics were consistent with those in CARTITUDE-1.13 Mean Cmax and AUC0-28d were slightly higher in the BsAb group than the ADC group, as were peak cytokine levels. These observations may be related to the increased proportion of T cells with an effector memory phenotype at apheresis. However, given the small sample size and large interindividual variability, these trends should be interpreted with caution. Serum BCMA decreased post infusion, indicating anti-BCMA activity of CAR-positive T cells. Baseline serum BCMA did not predict response to cilta-cel.
Although this study indicates that patients who have received BCMA-targeted therapy can respond to cilta-cel, the small sample size, as well as the heterogeneity of the type, duration, and timing of prior anti-BCMA therapy, are limitations. An additional limitation is that 10 (50%) patients were not assessable for the primary end point, mostly due to bone marrow hypocellularity and consequent inability to identify the baseline clone by next-generation sequencing. Furthermore, advanced patients with extramedullary disease may have diffuse marrow involvement or no infiltration. Response assessment in the BsAb-exposed group was limited, as 2 patients died before confirmation of response. Further, because of the low patient numbers, this study did not examine responses for patients who were exposed vs refractory to anti-BCMA therapy before cilta-cel treatment. Finally, prior anti-BCMA treatment typically occurred under other study protocols from different sponsors, so details surrounding those treatments (including dosing) were unavailable. These caveats make it difficult to draw firm conclusions regarding the optimal sequencing of these agents before initiating cilta-cel. Further prospective studies and real-world retrospective clinical experience are needed for a better understanding of sequencing BCMA-targeting therapies.
In summary, heavily pretreated patients with RRMM who have been previously exposed to a noncellular BCMA-targeting agent can respond to cilta-cel, with 60% of patients achieving a response. An increased understanding of the best candidates for cilta-cel treatment after other BCMA-targeting therapy will be key to optimizing outcomes. These initial results, along with continued follow-up of this cohort, inform sequencing of BCMA-targeted agents to maximize patient benefit.
Acknowledgments
The authors thank the patients who volunteered to participate in the study, their families and caregivers, the physicians and nurses who cared for the patients and supported this clinical trial, staff members who enrolled patients, and staff members involved in data collection and analysis. They also thank Venn Life Sciences ED BV, Netherlands, for their support of data activities throughout this study. Medical writing support was provided by Julie Nowicki of Eloquent Scientific Solutions.
This work was supported by Janssen Research and Development and Legend Biotech USA Inc. Medical writing support was funded by Janssen Global Services.
Authorship
Contribution: all authors contributed to the study design, study conduct, and data analysis and interpretation and participated in drafting and revising the manuscript and approved the final version for submission; and J.S.-M. had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Conflict-of-interest disclosure: A.D.C. received research funding from GlaxoSmithKline and Novartis and served as a consultant or advisor for AstraZeneca, Bristol Myers Squibb/Celgene, Genentech/Roche, GlaxoSmithKline, Janssen, Oncopeptides, and Takeda. M.-V.M. received honoraria from or served on the board of directors or on an advisory committee for Amgen, Adaptive Biotechnologies, Bristol Myers Squibb/Celgene, Janssen, Oncopeptides, Pfizer, Regeneron, Roche, Sanofi, Sea-Gen, and Takeda. Y.C.C. received research funding from Amgen, Karyopharm, and Takeda and received honoraria or served as a consultant or advisor to Amgen, GSK, Janssen, Neopharm/Promedico, and Takeda. P.R.-O. received honoraria from or served on speaker bureaus for Bristol Myers Squibb/Celgene, Janssen, Amgen, GlaxoSmithKline, Sanofi, Regeneron, and Oncopeptides. B.P. received research funding from Bristol Myers Squibb/Celgene, Roche, and Sanofi and served as a consultant or advisor for Adaptive, Amgen, Bristol Myers Squibb, Celgene, GlaxoSmithKline, Janssen, Kite, Roche, Sanofi, and Takeda. N.W.C.J.v.d.D. received research funding from Janssen Pharmaceuticals, Amgen, Celgene, Novartis, Cellectis, and Bristol Myers Squibb and served as a consultant or advisor to Janssen, Amgen, Celgene, Bristol Myers Squibb, Takeda, Roche, Novartis, Bayer, Adaptive, and Servier. T.M. received research funding from Janssen. A.S. received research funding from Bristol Myers Squibb, Janssen, GlaxoSmithKline, Regeneron, and Sutro and served as a consultant or advisor to Bristol Myers Squibb, Janssen, and GlaxoSmithKline. K.C.D.B., C.C., J.M.S., H.V., W.D., L.W., M.V., T.R., X.X., P.M., and E.Z. are employed by Janssen. M.A., T.N., and L.P. are employed by Legend Biotech USA Inc. I.A. has received honoraria from or served as a consultant or advisor to GlaxoSmithKline, Janssen, Neopharm/Promedico, Medison, and Gilead. J.S.-M. has served as a consultant, member of board of directors, or on advisory committees for AbbVie, Amgen, Bristol Myers Squibb/Celgene, GlaxoSmithKline, Janssen, Karyopharm, Merck Sharp & Dohme, Novartis, Regeneron, Roche, Sanofi, Secura Bio, and Takeda.
Correspondence: Jesus San-Miguel, Clinica University of Navarra, Avda PIO XII, 36 31008, Pamplona, Navarra, Spain; e-mail: sanmiguel@unav.es.
References
Author notes
The data sharing policy of Janssen Pharmaceutical Companies of Johnson & Johnson is available at https://www.janssen.com/clinical-trials/transparency. As noted on this site, requests for access to the study data can be submitted through the Yale Open Data Access Project site at http://yoda.yale.edu.
The online version of this article contains a data supplement.
There is a Blood Commentary on this article in this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.