TO THE EDITOR:
The assembly of canonical and noncanonical inflammasomes activates caspase-1 and caspase-11, respectively, culminating in the cleavage of a common downstream effector, gasdermin D (GSDMD). GSDMD forms membrane pores and consequently triggers a form of lytic cell death termed pyroptosis.1-5 GSDMD belongs to the gasdermin family of proteins along with the following 5 other paralogs: GSDMA, GSDMB, GSDMC, GSDME, and DFNB59. GSDMD is the most widely studied protein in the gasdermin family, and its regulation of lytic cell death has firmly implicated it in the pathogenesis of several inflammatory diseases, including sepsis. Recent studies have shown that GSDMD regulates host defenses and contributes to survival in both lipopolysaccharide6-9 and cecal ligation and puncture (CLP)-induced sepsis models.10,11 In patients with sepsis, biomarkers of GSDMD activation correlate with disseminated intravascular coagulation scores, and its deletion in mice is shown to reduce endotoxemia-induced disseminated intravascular coagulation,12 confirming the detrimental effect of GSDMD in sepsis.
GSDMD’s contribution to sepsis is generally thought to be related to GSDMD-mediated macrophage pyroptosis.6-8,10,11 However, GSDMD is also expressed in neutrophils, another key innate immune cell regulating acute infection and inflammation. GSDMD is usually cleaved by inflammatory caspases -1, -4, -5, -8, and -11 in monocytes and macrophages.13,14 However, in neutrophils, GSDMD is cleaved by caspase-11,15 neutrophil elastase ,15,16 and cathepsin G,17 suggesting differential physiological regulation of GSDMD in different myeloid cells. Neutrophil elastase-induced cleavage of GSDMD in neutrophils during Escherichia coli infection causes pyroptosis, thereby reducing neutrophil accumulation in the peritoneal cavity and hindering bacterial clearance.15 GSDMD also plays a vital role in the generation of neutrophil extracellular traps (NETs),16,18 which are extracellular DNA fibers networked with histone and cytoplasmic granule proteins that entangle and contain bacteria and are also implicated in thrombosis, atherosclerosis, autoimmune diseases, and sepsis.19-21
In a recent study in Blood, Silva et al reported that GSDMD regulates sepsis by promoting NET formation in a mouse polymicrobial sepsis model.22 Using Gsdmd whole-body knockout (Gsdmd KO) mice, they showed that these mice produce fewer NETs and reduced levels of inflammatory cytokines, including IL-1β and TNF-α, ultimately improving the survival in CLP-induced sepsis. These results are consistent with other studies showing that GSDMD is detrimental during sepsis10,11 and suggest that in neutrophils, it plays a critical role in the pathogenesis of sepsis. However, a key experiment missing from this study was to use neutrophil-specific conditional knockout mice to definitively confirm the causal role of neutrophil GSDMD in sepsis. Therefore, we generated neutrophil-specific conditional Gsdmd KO mice (Gsdmdflox/flox MRP8-Cre+) (supplemental Figure 1A; available on the Blood website). MRP8creTg is a transgenic mouse line generated to specifically express Cre-recombinase and enhanced green fluorescent protein in neutrophils and is widely used to study neutrophil-specific function in pathobiological conditions.23,24 The specific deletion of neutrophil Gsdmd was confirmed by western blotting (supplemental Figure 1B-F) and genotyping (supplemental Figure 1G-H). Gsdmdflox/flox MRP8-Cre+ mice were similar to their wild-type (WT) littermate controls in appearance, behavior, neutrophil maturation subsets (supplemental Figure 2A), and total neutrophil count in the bone marrow (supplemental Figure 2B) and peripheral blood (supplemental Figure 2C). Neutrophil morphology was also normal in the GSDMD conditional KO mice (supplemental Figure 2D). Neutrophils isolated from the neutrophil-specific conditional Gsdmd KO mice displayed normal neutrophil functions such as respiratory burst (supplemental Figure3A) and phagocytosis (supplemental Figure 3B). In addition, GSDMD deletion did not affect the expression of NLRP3, caspase-11, or elastase in both unchallenged and bacteria-challenged neutrophils (supplemental Figure 4).
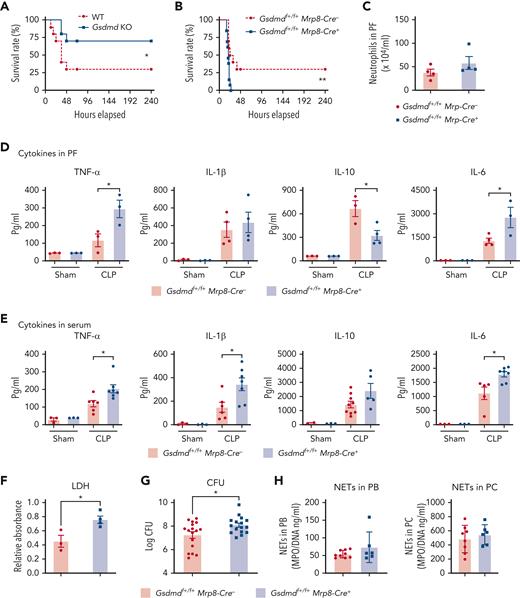
CLP was used to induce sepsis in Gsdmdflox/flox MRP8-Cre+ mice and their littermate controls (Gsdmdflox/flox MRP8-Cre−), and their survival was observed. Whole-body Gsdmd KO mice were included for comparison. Consistent with previous reports,6,10 whole-body Gsdmd KO mice did not die from CLP-induced polymicrobial sepsis compared with their WT counterparts (Figure 1A). Because Silva et al22 showed that GSDMD increases the severity of sepsis by inducing NETs, we expected a protective phenotype in Gsdmdflox/flox MRP8-Cre+ mice but, surprisingly, this was not the case (Figure 1B), suggesting that neutrophil GSDMD is not detrimental in sepsis. Instead, sepsis was more severe in Gsdmdflox/flox MRP8-Cre+ mice (Figure 1B).
Disruption of neutrophil GSDMD does not protect the host from sepsis. (A) Polymicrobial sepsis was induced in WT and Gsdmd KO mice (N = 10/group) via CLP, and survival was monitored for up to 10 days. (B) Survival of neutrophil-specific–conditional KO mice and their counterparts during sepsis. Gsdmdflox/floxMRP8-Cre+ (N = 13) and WT counterpart mice (N = 10) were subjected to polymicrobial sepsis using CLP. The survival was monitored. Data were analyzed using the log-rank (Mantel-Cox) test. (C-H) Sepsis was induced in Gsdmdf+/f+MRP8-Cre+ and littermate WT control Gsdmdf+/f+MRP8-Cre- mice via CLP. Twelve hours after CLP, mice were euthanized to collect PF and serum. (C) Neutrophil recruitment to the peritoneal cavity as measured by flow cytometry. (D) Cytokines (TNF-α, IL-1β, IL-10, and IL-6) in cell-free PF. (E) Cytokines in serum. (F) Lactate dehydrogenase levels in PF. (G) The bacterial burden in the PF was quantified. (H) NETs production in peripheral blood and peritoneal cavity. Data are presented as mean ± SEM. The log-rank (Mantel-Cox) test was used to analyze survival differences between groups. In other graphs, t-test was used to compare the means. ∗P < .05; ∗∗P < .01. CFU, colony forming units; LDH, lactate dehydrogenase; PC, peritoneal cavity; PF, peritoneal lavage fluid; SEM, standard error of the mean.
Disruption of neutrophil GSDMD does not protect the host from sepsis. (A) Polymicrobial sepsis was induced in WT and Gsdmd KO mice (N = 10/group) via CLP, and survival was monitored for up to 10 days. (B) Survival of neutrophil-specific–conditional KO mice and their counterparts during sepsis. Gsdmdflox/floxMRP8-Cre+ (N = 13) and WT counterpart mice (N = 10) were subjected to polymicrobial sepsis using CLP. The survival was monitored. Data were analyzed using the log-rank (Mantel-Cox) test. (C-H) Sepsis was induced in Gsdmdf+/f+MRP8-Cre+ and littermate WT control Gsdmdf+/f+MRP8-Cre- mice via CLP. Twelve hours after CLP, mice were euthanized to collect PF and serum. (C) Neutrophil recruitment to the peritoneal cavity as measured by flow cytometry. (D) Cytokines (TNF-α, IL-1β, IL-10, and IL-6) in cell-free PF. (E) Cytokines in serum. (F) Lactate dehydrogenase levels in PF. (G) The bacterial burden in the PF was quantified. (H) NETs production in peripheral blood and peritoneal cavity. Data are presented as mean ± SEM. The log-rank (Mantel-Cox) test was used to analyze survival differences between groups. In other graphs, t-test was used to compare the means. ∗P < .05; ∗∗P < .01. CFU, colony forming units; LDH, lactate dehydrogenase; PC, peritoneal cavity; PF, peritoneal lavage fluid; SEM, standard error of the mean.
To investigate if the increased susceptibility of Gsdmdflox/flox MRP8-Cre+ mice to sepsis was because of hyperinflammation, we analyzed neutrophil recruitment and inflammatory cytokine levels in peritoneal lavage fluid collected from septic mice. Although there was no significant difference in neutrophil numbers in the peritoneal cavity between the groups (Figure 1C), Gsdmdflox/flox MRP8-Cre+ mice had higher levels of proinflammatory cytokine, TNF-α, and IL-6, and lower levels of anti-inflammatory cytokine IL-10 in their peritoneal fluid than littermate controls (Figure 1D). Serum collected from septic Gsdmdflox/flox MRP8-Cre+ mice also contained higher levels of proinflammatory cytokines, such as TNF-α, IL-1β, and IL-6 (Figure 1E), confirming excessive inflammation in these mice compared with the WT littermates. In addition, Lactate dehydrogenase, a marker of inflammatory cell death, was also elevated in the peritoneal lavage fluid collected from Gsdmdflox/flox MRP8-Cre+ mice (Figure 1F). Notably, Gsdmdflox/flox MRP8-Cre+ mice showed higher bacterial burden than the WT littermates (Figure 1G). Finally, although GSDMD is required for NET formation in an in vitro setting,16,18 CLP-induced–NET formation was not significantly altered in neutrophil-specific–conditional KO mice, suggesting that the hyperinflammation or defective bactericidal activity in these mice was unlikely to be mediated by NETs (Figure 1H). A recent study25 showed that neutrophil NET formation can be indirectly regulated by GSDMD in other cell types. In CLP-induced sepsis, GSDMD triggers platelet pyroptosis, leading to the release of oxidized mitochondrial DNA (ox-mtDNA), which in turn promotes NET formation.25 Thus, it provides an alternative mechanism for GSDMD-mediated–NET formation, and a potential explanation for the unaltered in vivo NET formation in the neutrophil-specific–conditional KO mice.
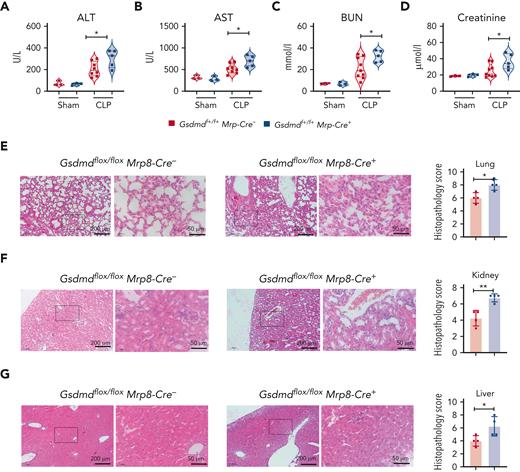
To evaluate the extent of multiorgan dysfunction in these mice, circulating levels of alanine transaminase, aspartate aminotransferase, blood urea nitrogen, and creatinine were evaluated. Consistent with survival, Gsdmdflox/flox MRP8-Cre+ mice had high levels of serum alanine transaminase (Figure 2A), aspartate transaminase (Figure 2B), blood urea nitrogen (Figure 2C), and creatinine (Figure 2D). Furthermore, the semiquantitative histopathological examination of Gsdmdflox/flox MRP8-Cre+ mice’s lungs (Figure 2E), kidneys (Figure 2F), and liver (Figure 2G) revealed more tissue damage than littermate WT controls. Taken together, these findings suggest that the deletion of neutrophil GSDMD does not protect the host from polymicrobial sepsis, instead it triggers even more severe sepsis, contrary to the conclusions reported by Silva et al.22
GSDMD deficiency in neutrophils increases multiorgan dysfunction during lethal sepsis. (A-G) Polymicrobial sepsis was induced in Gsdmdf+/f+MRP8-Cre+ and littermate WT control (Gsdmdf+/f+MRP8-Cre−) mice via CLP. Twelve hours after CLP, mice were euthanized to collect serum, lungs, kidneys, and liver. ALT (A), AST (B), BUN (C), and creatinine (D) were measured in the serum. Histopathological images of the lungs (E), kidneys (F), and liver (G) and their corresponding scores. t-test was used to compare the means between 2 groups. ∗P < .05; ∗∗P < .01. ALT, alanine transaminase; AST, aspartate aminotransferase; BUN, blood urea nitrogen.
GSDMD deficiency in neutrophils increases multiorgan dysfunction during lethal sepsis. (A-G) Polymicrobial sepsis was induced in Gsdmdf+/f+MRP8-Cre+ and littermate WT control (Gsdmdf+/f+MRP8-Cre−) mice via CLP. Twelve hours after CLP, mice were euthanized to collect serum, lungs, kidneys, and liver. ALT (A), AST (B), BUN (C), and creatinine (D) were measured in the serum. Histopathological images of the lungs (E), kidneys (F), and liver (G) and their corresponding scores. t-test was used to compare the means between 2 groups. ∗P < .05; ∗∗P < .01. ALT, alanine transaminase; AST, aspartate aminotransferase; BUN, blood urea nitrogen.
Silva et al22 transferred WT neutrophils or monocytes into Gsdmd KO mice and found that adoptive transfer of WT neutrophils but not monocytes increased organ dysfunction and NET formation during sepsis. Therefore, they inferred that neutrophil GSDMD causes organ dysfunction by increasing NETs and inflammation. However, neutrophil transfusion may itself cause severe organ damage independent of the GSDMD expression. The authors did not include Gsdmd KO neutrophils as a key control, which may have revealed that they too trigger severe sepsis. This is important because adding neutrophils during sepsis could further increase the severity of inflammation and organ dysfunction. Excessive neutrophil recruitment and activation can cause tissue damage and lead to multiorgan damage,26 whereas removing neutrophils from the circulation using leucodepletion filters in patients with sepsis significantly improves pulmonary, myocardial, and renal function,27,28 although 1 of the studies showed that the hospital outcomes were similar in terms of mortality and length of stay.28
In conclusion, our results reveal a new and unexpected role of neutrophil GSDMD in sepsis. By using neutrophil-specific conditional Gsdmd KO mice to clarify the function of neutrophil GSDMD in sepsis, we show that though whole-body deletion of GSDMD protects against sepsis, its specific deletion from neutrophils not only fails to protect against sepsis but may even make it worse.
Currently, the mechanism that leads to hyperinflammation in neutrophil-specific KO mice is still largely unknown. Although neutrophils are the most abundant cells in the peripheral blood during sepsis, other myeloid cells such as macrophages, monocytes, and dendritic cells are considered a major source of proinflammatory cytokines in many experimental models, including sepsis.29,30 GSDMD is required for pyroptosis and IL1β secretion, thus cytokine production in GSDMD whole-body KO mice is diminished during sepsis.3,31,32 In contrast, we detected higher cytokine production in peritoneal lavage fluid collected from CLP-challenged neutrophil-specific GSDMD KO mice than from WT mice. CLP-induced polymicrobial infection and the subsequent exacerbated immune response is triggered by the microbes released from the cecum into the peritoneal cavity. The gut microbiota consists of diverse forms of microorganisms, including some intracellular microbes.33 In the CLP model, a higher bacterial load was detected in neutrophil-specific GSDMD conditional KO mice (Figure 1G), suggesting that neutrophil GSDMD is important for clearing certain gut bacteria. GSDMD depletion in neutrophils may compromise their bactericidal activity and lead to more severe inflammation and organ damage. Pyroptotic cell death works as a key host defense mechanism against intracellular pathogens by removing their replication niche and making them susceptible to phagocytosis and killing by secondary phagocytes.34 Although neutrophils are best known for their phagocytic and microbicidal properties in extracellular infections, recent studies indicate that they also play a critical role in host defense against certain intracellular pathogens,35 such as Listeria monocytogenes36 and Legionella pneumophila.37 GSDMD-mediated pyroptosis might be important for not only macrophage- but also neutrophil-mediated clearance of intracellular microbes. In neutrophil-specific conditional Gsdmd KO mice, the impaired bacterial killing capability may cause more severe infection, which in turn leads to elevated cytokine production by myeloid cells other than neutrophils, leading to hyperinflammation in CLP-challenged mice.
Acknowledgment
The authors thank their lab member Rong Xu for her helpful discussion.
This work was supported National Institutes of Health (NIH), National Heart, Lung, and Blood Institute (NHLBI) grant NIH P01 HL095489, Molecular Mechanisms of Blood Cell Transfusion (Project 3); NIH, National Institute of Allergy and Infectious Diseases (NIAID) grant NIH R01AI141386, Cell-Based High-throughput chemical genetic screening for GSDMD Inhibitors; NIH, NHLBI grant NIH R01HL092020, Enhancing neutrophil function in neutropenia-related pneumonia; NIH, NIAID grants NIH R01AI142642, Regulation of IL-1β bioactivity by cysteine S-glutathionylation and NIH R01 AI145274, Regulation of neutrophil death by GSDMD in Candida albicans infection; Tianjin Municipal Science and Technology Commission Grant: 21JCQNJC01550; and Haihe Laboratory of Cell Ecosystem Innovation Fund: HH22KYZX0019.
Authorship
Contribution: L.G., F.L., F.M., and H.R.L. conceptualized and designed the experiment; L.G., F.L., A.Y.H., A.B., H.Y., and Z.Z. executed the experiments; F.L. and L.G. analyzed the data; L.G. drafted the manuscript; and H.R.L. revised and finalized the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Hongbo R. Luo, Department of Laboratory Medicine, Boston Children's Hospital, Enders Research Bldg, Room 811, 320 Longwood Ave, Boston, MA 02115; e-mail: hongbo.luo@childrens.harvard.edu.
References
Author notes
Data are available on request from the corresponding author, Hongbo R. Luo (hongbo.luo@childrens.harvard.edu).
The online version of this article contains a data supplement.
There is a Blood Commentary on this article in this issue.
F.L. and L.G. contributed equally to this study.