Introduction:
Mastocytosis is a spectrum of diseases characterized by the accumulation of atypical mast cells (MC) in tissues. The most frequent form in adult patients is systemic mastocytosis (SM) including indolent SM, bone marrow mastocytosis, smoldering SM and advanced SM (adv-SM). Adv-SM encompasses aggressive SM (ASM), MC leukemia (MCL), SM associated with hematological neoplasms (SM-AHN) and is associated with a poor outcome. Prior the use of tyrosine kinase inhibitors (TKIs), therapeutic options were limited in adv-SM patients. The first TKI developed for adv-SM with KIT D816V mutation was midostaurin, which greatly improved outcomes for patients. However, most patients are primary refractory or relapse early (<12 months) after midostaurin onset. To identify high risk adv-SM patients, a prognostic score the so-called Mutation-Adjusted Risk Score (MARS) have been developed. However, given the heterogeneity of outcomes according to subtype of adv-SM, the identification of high-risk patients within each subtype is critical to optimize therapy. To address this question, we investigated predictive value on outcome of MARS score risk in midostaurin treated Adv-SM patients in a larger group of patients, according to each subtype of adv-SM.
Methods:
We conducted a nationwide retrospective study of all patients with Adv-SM patients included in the French national mastocytosis reference center registry (CEREMAST). Main inclusion criteria were: (i) adults patients treated with midostaurin in France since 2009 (ii) diagnosis of adv-SM according to WHO 2016 classification (iii) presence of C findings. Patients that have received combination therapy (i.e. azacytidine) or with chronic type of MCL without C finding or lymphoid neoplasm AHN or AML prior midostaurin onset were excluded. Response to midostaurin was assessed according to Valent criteria and MARS score was used to stratify patients into 3 risk groups (high risk, intermediate and low).
Results:
Overall, 170 patients treated with midostaurin were identified, including 46 ASM patients, 11 MCL patients and 113 SM-AHN patients. Among SM-AHN patients, chronic myelomonocytic leukemia was the most frequent neoplasm (52%). Response to midostaurin was significantly different regarding subtype of adv-SM (73% in ASM vs 27% in MCL vs 50% in SM-AHN, p=0.006). With a median follow-up of 19 months since midostaurin start, median overall survival (OS) was 69, 9.9 and 32 months (p=0.0013) and time to treatment failure (TTF) was 30, and 3.6 and 9 months in ASM, MCL and SM-AHN (p<0.0001), respectively.
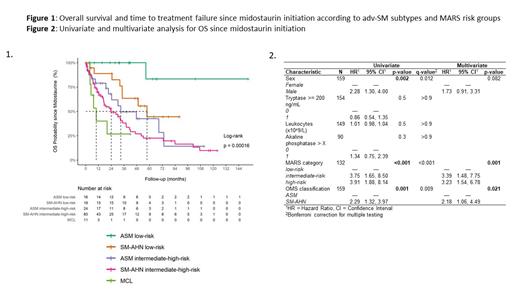
MARS risk group distribution was heterogeneous between ASM, MCL and SM-AHN (p=0.026). In MARS low risk group, median OS was not reached (NR) and 58 months in ASM and SM-AHN respectively (p=0.034, Figure 1). Median OS for both low risk ASM and low risk SM-AHN were significantly different compared with intermediate/high-risk (int/high-risk) groups (NR vs 33 months for ASM, p=0.003 and 58 months vs 24 months, p=0.017). Int/high-risk SM-AHN was the only group with a median OS not significantly different compared with MCL (24 months vs 9 months p=0.266)
Median TTF was 60 months for low risk ASM, 26 months for low risk SM-AHN and only 6.7 months for high/int-risk ASM, 8.4 months for high/int-risk SM-AHN and 3.6 months for MCL.
For the 19 patients who underwent hematopoietic stem cell transplantation (HSCT), median OS was 88 months but did not reach statistical significance compared to non-transplanted patients (47 months, p=0.15). In multivariate analysis, MARS and WHO mastocytosis subtype were the only variables independently associated with OS ( figure 2). MARS and midostaurin response were not predictive for both AHN progression and AML transformation in SM-AHN, which occurred in 31% and, 22% of patients respectively. In contrast, presence of portal hypertension/ascites (68% vs 29%, p<0.001) and abnormal karyotype (40 vs 11%, p=0.007) were predictive of AML transformation.
Conclusions:
We report herein the largest cohort of adv-SM patients treated with midostaurin. We identified portal hypertension/ascites and karyotype abnormalities as risk factors predictive of AML evolution. Subtype of adv-SM are heterogeneous and, together with MARS, are predictive for both OS and TTF. According to these variables, 5 sub-groups of adv-SM have been identified with different outcomes prompting for specific management.
Disclosures
Heiblig:Jazz Pharmaceuticals: Honoraria; AbbVie: Honoraria; Pfizer Inc.: Honoraria; Astellas: Honoraria; Servier: Honoraria. Livideanu:Lilly, Novartis, UCB: Consultancy; Blueprint Medicines Corporation: Membership on an entity's Board of Directors or advisory committees; ABScience: Membership on an entity's Board of Directors or advisory committees; Novartis: Research Funding. Barete:Blueprint Medicines Corporation: Consultancy, Other: Fees for symposium and scientific boards; financial support for clinical trials; AbbVie: Other: Fees; Leo Pharma Laboratories: Other: Fees. Damaj:Takeda, Blueprint Medicines Corporation, and Thermo Fisher: Consultancy, Other: Advisory Role; Takeda, AbbVie and Pfizer: Other: Travel and Accommodation Expenses. Frenzel:CSL Berhing: Consultancy, Research Funding; Biomarin: Consultancy; Roche: Consultancy; Pfizer: Consultancy, Other: Grant, Research Funding. Molina:Janssen: Other: Travel and congress fees. Zunic:Abbvie, Janssen, Sanofi: Other: Travel Support. Viallard:EUSAPHARMA: Consultancy. Hermine:AB Science: Consultancy, Other: Shareholder; AB Science, BMS/Celgene, Alexion, Novartis, and Inatherys: Research Funding.