BACKGROUND: The development of targeted immunotherapy for acute myeloid leukemia (AML) is challenging due to the clonal heterogeneity of the disease and the lack of single AML-specific target antigens. MP0533 is a multi-specific, half-life extended CD3-engaging DARPin (Designed Ankyrin Repeat Protein). It simultaneously targets CD33, CD123, and CD70, thereby enabling avidity-driven T cell-mediated killing of leukemic stem cells (LSCs) and malignant blast cells known to co-express these targets. MP0533's affinity to each target is tuned to preferentially kill malignant cells which co-express at least two of these three antigens, while preserving a therapeutic window towards healthy cells. MP0533 was shown to induce a tumor-specific endogenous T-cell response in ex vivo samples from AML patients and led to T-cell-mediated eradication of tumors in AML xenograft mouse models, with limited cytokine release or other serious adverse reactions (Bianchi et al., Blood 2022;140[Suppl 1]:2251-2). Herein, we present early observations of the first three MP0533 dose-escalation regimens (DR) of the ongoing first-in-human, multicenter, single-arm, open-label, phase 1/2a study.
METHODS: The objectives of this ongoing trial are to evaluate the safety/tolerability, pharmacokinetics (PK), and pharmacodynamics, as well as preliminary antileukemic activity of MP0533 monotherapy in patients with relapsed/refractory (R/R) AML or myelodysplastic syndrome (MDS)/AML (NCT05673057). Patients with an Eastern Cooperative Oncology Group (ECOG) performance status of 0 to 2, and a life expectancy of ≥12 weeks are eligible. Up to 45 patients will be enrolled in the dose-escalation part of the trial. Patients receive MP0533 once per week starting at day 15 after a stepwise dose increase on days 1, 5, and 8, and until disease progression or unacceptable toxicity. One cycle corresponds to 28 days. DR escalation of MP0533 is guided by two Bayesian logistic regression models estimating the joint probability of cytokine release syndrome (CRS) and non-CRS dose-limiting toxicities (DLTs). Bone marrow examinations are performed at weeks 4, 8, and 12. Responses are determined using 2022 European LeukemiaNet (ELN) criteria with an assessment of hematologic improvement. Treatment-emergent adverse events (TEAEs) are assessed according to National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) v5.0.
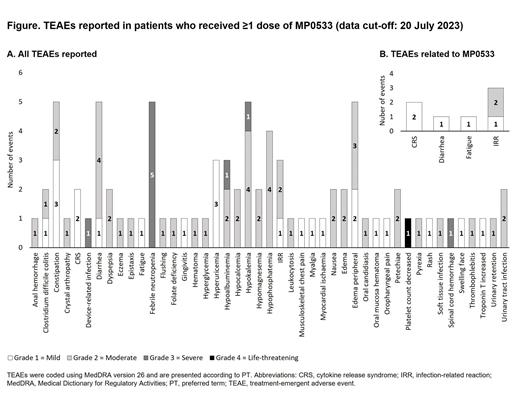
RESULTS: As of data cut-off (20 July 2023), 5 patients received ≥5 infusions of MP0533 across 3 DRs preceded by step-up dosing (1 patient each at DR 1 and DR 2, respectively, and 3 patients at DR 3), including 2 women and 3 men. The median age at enrollment was 79 years (range 68-81). All patients had adverse risk mutations. Patients received a median number of 1 prior antileukemic therapy line (range 1-2) and on average 2 cycles of MP0533 treatment (range 1-3). To date, one patient with a TP53 mutation achieved a morphologic leukemia-free state after 8 weeks and complete remission with incomplete hematologic recovery (CRi) after 12 weeks of treatment with DR 3. No DLTs have been observed in any DR so far. TEAEs considered to be related to MP0533 were Grade 2 infusion-related reaction (2 patients), Grade 1 CRS (2 patients), and Grade 1 diarrhea and fatigue (1 patient). No Grade ≥3 drug-related TEAEs were reported. The reported TEAEs (Figure) reflected the patients' disease course. Preliminary PK results up to DR 3 confirmed that all patients were exposed to MP0533 and exposure levels were broadly aligned with PK/PD model predictions of MP0533 in serum.
CONCLUSION: The results of this ongoing phase 1/2a study indicated an acceptable safety profile of MP0533 monotherapy in 5 patients up to DR 3 with weekly infusions. Preliminary response data are encouraging, with one response observed in 1 of 2 patients evaluable for response in DR 3 to date.
Disclosures
Jongen-Lavrencic:Ellipses Pharma: Research Funding. de Leeuw:Abbvie: Consultancy, Other: Advisory role ; Abbvie: Other: Travel, accomodation expenses; Roche: Consultancy, Other: Advisory role; Servier: Consultancy, Other: Advisory role; Takeda: Consultancy, Other: Advisory role. Dymkowska:Cantargia: Consultancy; Morphosys: Consultancy; Debiopharm: Consultancy. Subklewe:Janssen: Consultancy, Honoraria, Research Funding, Speakers Bureau; Roche: Consultancy, Honoraria, Other: Travel Support, Research Funding, Speakers Bureau; BMS/Celgene: Consultancy, Honoraria, Research Funding, Speakers Bureau; Amgen: Consultancy, Honoraria, Research Funding; Miltenyi Biotec: Consultancy, Honoraria, Research Funding; Novartis: Consultancy, Honoraria, Research Funding, Speakers Bureau; Gilead/Kite: Consultancy, Honoraria, Other: Travel Support, Research Funding, Speakers Bureau; Pfizer: Consultancy, Honoraria, Other: Travel Support, Speakers Bureau; AstraZeneca: Speakers Bureau; Seagen: Research Funding; Takeda: Consultancy, Honoraria, Research Funding; Ichnos Sciences: Consultancy, Honoraria; AvenCell: Consultancy, Honoraria; Incyte Biosciences: Consultancy, Honoraria; Molecular Partners: Consultancy, Honoraria, Research Funding; GSK: Speakers Bureau; LAWG: Speakers Bureau; Springer Healthcare: Speakers Bureau; AbbVie: Consultancy, Honoraria; Autolus: Consultancy, Honoraria; advesya (CanCell Therapeutics): Consultancy, Honoraria; Genmab US: Consultancy, Honoraria; Interius BioTherapeutics: Consultancy, Honoraria; Nektar Therapeutics: Consultancy, Honoraria; Orbital Therapeutics: Consultancy, Honoraria; Sanofi: Consultancy, Honoraria; Scare: Consultancy, Honoraria.