Background
Autologous hematopoietic stem cell transplantation (ASCT) and chimeric antigen receptor T‐cell therapy (CART) are salvage therapies that are utilised for the treatment of relapsed or refractory (R/R) B-cell lymphoma. However, whether the combination therapy of ASCT and CART (ASCT‐CART) can improve the survival of R/R B-cell lymphoma remains unknown.
A im
The objective is to explore the effectiveness and safety of ASCT‐CART in the treatment of refractory/relapsed B-cell lymphoma and compare the effects of disease status before ASCT-CART on patient survival.
Methods
From October 2019 to October 2022, 49 patients with R/R B-cell lymphoma tract were enrolled. The male/female ratio was 1.2:1, with a median age of 40 years (range, 16-69 years).The diagnosis included diffuse large B-cell lymphoma (DLBCL) (n=31), High-grade B-cell lymphoma(n=8), Primary mediastinal B-cell lymphoma (PMBCL) (n=7), Burkitt lymphoma (BL) (n=2) and Richer (n=1). Among them , stage III-IV accounted for 43/49 (87.8%) and GCB subtype 22/49 (44.9%),and received a median of 7 cycles(3-14)of prior treatment, previous radiotherapy was 8/49 (16.3%), and previous other target CART patients were 20/49 (40.8%).The disease status of patients before ASCT-CART was 23 patients in complete remission, 17 patients in partial remission, 3 patients with stable disease, and 6 patients with progressive disease.Patients were treated with BEAM and fludarabine before ASCT. Autologous stem cells were transfused with D0, and the median number of CD34+ cells was 4.14*106/kg(range, 0.7-12.99*106/kg). CART cells were transfused on D1, and the median infusion of CART cells was 2.06*106/kg(range, 0.07-9.5*106/kg).
Results
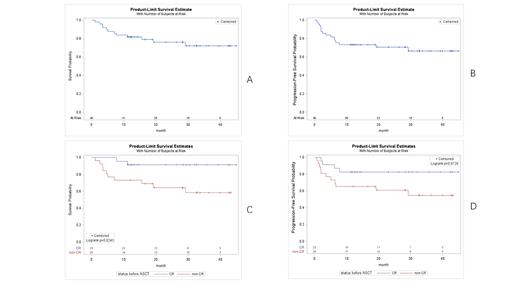
In efficacy assessment at 3 months after treatment, the objective response rate (ORR) was 40/49(81.6%), and the complete response rate (CRR) was 38/49 (77.6%).After a median follow-up of 23.1 months, the median overall survival (OS) was not reached, with 1-year OS being 81.6%, 2-year OS being 76%, and 3-year OS being 72.3%(Figure 1A). Median progression-free survival (PFS) was not reached, with 1-year PFS at 73.5%, 2-year FFS at 70.5%, and 3-year FFS at 66.6%(Figure 1B) . Neutrophil and platelet engraftment in all cases on median days 16 (range 10-62 days) and 16 (range 7-52), respectively.Grade 3 or higher cytokine release syndrome (≥grade 3 CRS) and immune efector cell-associated neurologic toxicity syndrome (≥grade 3 ICANS) events occurred in 22.4%(11/49) and 4.1%(2/49)of the patients, respectively.In the 3-month efficacy evaluation of CR group before ASCT-CART treatment, ORR was 21/23(91.3%) and CRR was 20/23(87.0%). ORR and CRR were 19/26(73.1%) and 18/26 (69.2%) in patients who did not reach CR before ASCT-CART treatment.The 3-year OS and 3-year PFS were 91.3% and 82.6% in CR group before ASCT-CART treatment(Figure 1C,D). The 3-year OS and 3-year PFS in the non-CR group were 58.6% and 54.6% respectively(Figure 1C,D). Patients in CR group before ASCT-CART treatment had significantly prolonged OS and PFS (P=0.0243) and OS (P= 0.0720).
Summary/Conclusion
The ASCT-CART therapy could enhance its the remission rate and long-term efficacy in the treatment of advanced relapsed/refractory B-cell lymphoma with controllable safety. Patients who were in remission before ASCT-CART therapy achieved a longer term remission after ASCT-CART cell therapy.
Disclosures
No relevant conflicts of interest to declare.